This month’s blog features several recently reported small molecules with interesting structural motifs, including tetrahydroquinolines, spiroazetidines, diheterocyclic pyrroles, pyridinones, as well as covalent acrylamide electrophiles. In addition, these analogs were designed to bind a diverse list of molecular targets, such as SGRM, KRAS, CDK, NNRT, EGFR, ProRS, and SARS-CoV2.

B53

B53: Through an extensive SAR of 62 sulfonamide tetrahydroquinolines, B53 was identified as a selective glucocorticoid receptor modulator (SGRM) for utility in immunology. It was found to have potent transrepression (IC50 NF-κB = 9 nM) with no transactivation. The compound also reduces the expression of several inflammatory biomarkers such as IL-6, IL-1β, and TNF- α. In an in vivo DNCB-induced atopic dermatitis model, efficacy was observed at 20 mg/kg (P.O.)
Reference: https://doi.org/10.1021/acs.jmedchem.2c01082
TB-25
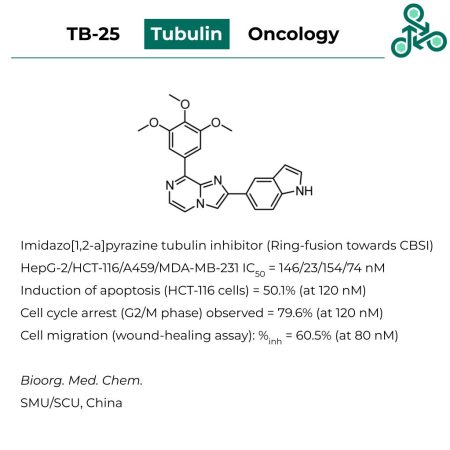
TB-25: Using a ring-fusion strategy towards a colchicine-binding site inhibiting pharmacophore, Imidazo[1,2-a]pyrazine TB-25 was identified as a tubulin small molecule inhibitor. Cytotoxicity was observed against HePG-2, HCT-116, A459, and MDA-MB-231 cells, with IC50 values of 146, 23, 154, and 74 nM, respectively. The induction of apoptosis in HCT-116 (colon cancer) cells was studied, with 50.1% induction at 120 nM. Cell cycle arrest (at G2/M phase) was also observed (79.6%, at 120 nM). In a wound-healing assay studying the relation between cell migration, and tubulin inhibition, 60.5% inhibition was observed at 80 nM of inhibitor.
Reference: https://doi.org/10.1016/j.bmc.2022.117098
Compound 15B

Compound 15B: Structurally derived from Spautin-1, Compound 15B is an EGFR inhibitor, targeting the mutant protein isoform (Ex19Del, L858R/T790M). From an original micromolar hit compound (5 μM potency) to Compound 15B (NSCLC PC-9 IC50 = 4 nM). Synthesis of the central quinazoline core involved formamide coupling via the use of 2-carboxyanilines. Upon co-incubation with established covalent EGFRi Afatinib, a 2x improvement in potency was observed (with the authors indicating a possible additive effect).
Reference: https://doi.org/10.1016/j.bmcl.2022.129066
B2
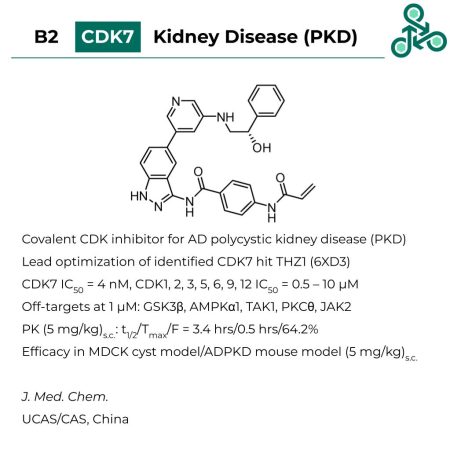
B2: Through lead optimization of a previously identified CDK7 targeting hit molecule (THZ1, PDB 6XD3), B2 was designed as a covalent CDK inhibitor, for utility in autosomal dominant polycystic kidney disease (AD-PKD). Potency was observed against CDK7 (IC50 = 4 nM), with less activity against other CDK isoforms (CDK1, 2, 3, 5, 6, 9, 12 IC50 = 0.5 – 10 μM). At 1 μM, a kinome screen revealed off targets as GSK3β, AMPKα1, TAK1, PKCθ, JAK2. Efficacy was observed in a MDCK cyst model/ADPKD mouse model (at 5 mg/kg) (S.C.).
Reference: https://doi.org/10.1021/acs.jmedchem.2c01334
JDQ443
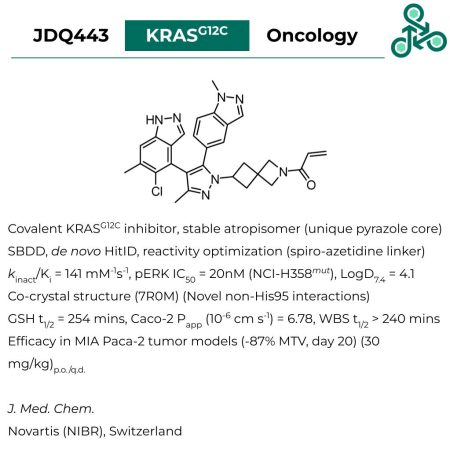
JDQ443: As a stable atropisomer (predicted interconversion barrier, ΔErot = 35.9 kcal mol-1) with a unique pyrazole core, JDQ443 was developed as a covalent KRASG12C inhibitor through a structure-based drug design strategy, coupled with de nova HitID, and electrophile reactivity optimization (via a spiro-azetidine linker). Activity against pERK was found (IC50 = 20 nM in NCI-H358mut), with a kinact/Ki value of 141 mM-1s-1. A co-crystal structure (7R0M) revealed the presence of novel interactions beyond canonical His95. Efficacy was demonstrated in a MIA Paca-2 tumor model (-87% MTV, day 20), at 30 mg/kg (p.o./q.d.).
Reference: https://doi.org/10.1021/acs.jmedchem.2c01438
Compound 9Q

Compound 9Q: As a N-pyridinylpyrimid-2-amine, this small molecule was identified as a dual inhibitor of CDK7/9 with noticeable % inhibition at 1 μM of 87, and 90%, respectively. This activity translated into an apparent Ki (CDK7H/CDK9T1) of 55, and 38 nM with [ATP] held at the enzyme Michaelis constant (Km). Broad cytotoxicity was observed in various cell lines, including MCF-7 (breast cancer), A2780 (ovarian cancer), H460 (non-small cell lung cancer) and MV4-11 (acute myeloid leukemia), with IC50 ranges between 150-550 nM. The induction of apoptosis was also established in MV4-11 cells, using FACS Annexin V/PI based protocols.
Reference: https://doi.org/10.1002/cmdc.202200582
Compound 1F

Compound 1F: This small molecule, with a bis-furyl-pyrrolo[3,4-b]pyridin5-one core was reported as a SARS-CoV2 inhibitor. Interestingly, the design/synthesis of this molecule (and related analogs) involved the use of a one-pot 3-component Ugi-Zhu reaction (UZ-3CR), in the presence of a Yb(OTf)3 catalyst, which generally involves the reaction of aldehydes, amines, and isocyanide motifs. The % inhibition of SARS-CoV2 infection Vero-E6 was found to be 31.9, and 15 % in EC1, EC2, respectively. The inhibitor was found to display both prophylactic, and therapeutic benefits. In a receptor-ligand docking study against MPro, NSP3, and the spike-glycoprotein, virtual binding scores of -5.71, -4.72, and -4.49 kcal mol-1 were observed, respectively.
Reference: https://doi.org/10.1039/D2MD00350C
Compound 3

Compound 3: This quinazoline molecule was identified as a Staphylococcus aureus (gram-positive bacteria) prolyl-tRNA synthetase (SaProRS) inhibitor. Interestingly, a co-crystal of the SaProRS: 3:AMPPNP ternary complex (PDB 5ZNK) suggested an ATP-aided mechanism of action. This was further supported by determining the Kd in the presence/absence of ATP, with values of 30 nM, and 376 μM (represents a ~104-fold increase). MIC values in E. coli (GPB)/MRSA252 (GNB) were found to be 1 μg/mL, with IC50 in SaProRS of 150 nM. Fluorescence-based thermal shift analysis against both SaProRS/ EcProRS found ΔTm (oC) values of 10.2, and 20.4 oC, respectively.
Reference: https://doi.org/10.1021/acs.jmedchem.2c01496
Compound 36A

Compound 36A: Using a structure-based drug design, and molecular docking strategy, 36A was identified as a non-nucleoside reverse transcriptase (NNRT) inhibitor, for anti-viral utility against HIV-1. Broad activity was observed against wild-type HIV-1 (EC50 = 2.2 nM), as well as several relevant mutants: L1001, K103N (EC50 = 3.0, 2.8 nM). Against RES056, an EC50 value of 53.3 nM was observed. Using SPR analysis, an HIV-1 Kd was found as 2.50 μM, which is comparable to clinical standard HIV-1 medicine Etravirine. Several pharmacokinetic parameters of the hydrochloride salt (36A.HCl) were also determined at 10 mg/kg (P.O.), including T1/2 = 5.12 hrs, Tmax = 0.25 hrs, and F (%) at 12%.
Reference: https://doi.org/10.1021/acs.jmedchem.2c00576