In this entry of Small Molecule Highlights, we bring together a journal-focused list of molecules, from the Journal of Medicinal Chemistry (J. Med. Chem.), highlighting various drug targets, such as EGFR, KIT, GPR88, PLK4, SERD, ATG, and STAT5. These analogs utilize several interesting heterocyclic aromatic scaffolds, including furanopyrimidines, imidazopyridines, pyrazolo[3,4-d]pyrimidines, furanylmethylene-2-pyrrolidinones, and benzothiophenes. Interestingly, one molecule utilizes a zwitterionic scaffold, whilst another uses potent, and selective targeted protein degradation to knockdown master regulator STAT5.

Compound 52

Compound 52: This furanopyrimidine was recently reported as a potent, and highly selective covalent inhibitor of EGFRL858R/T790M double-mutant, targeting Cys797 via the canonical acrylamide electrophile, and a 1,4-conjugate addition MOA. Inhibitor design utilized a structure-based strategy focused on scaffold-hopping and a structure-activity relationship based on varying the position of solubilizing groups between two core rings: Ring A/B. This analog utilizes a dimethylamino- group. Interestingly, several well-established covalent kinase inhibitors utilize basic 3o amines in this manner (e.g. Afatinib, Neratinib, Dacomitinib). Recent literature has suggested that beyond solubility, these basic motifs may behave as internal bases, promoting Cys-SH thiolate formation and subsequent covalent attack. In vitro, this inhibitor targets EGFRWT with an IC50 of 25 nM, while EGFRL858R/T790M with an IC50 of 15 nM. In cells, against A431 (EGFRWT), a CC50 of 1373 nM is reported, while in H1975 (EGFRMut), a value of 179 nM is found (>7x FS). Using a kinome screen, an S10 score of 0.017 was found. In rats (20 mg/kg), oral bioavailability (% F) of 26.8% was observed (p.o.). Finally, in vivo efficacy was reported in two xenograft models (BaF3/H1975), with TGI values of 74.9, and 97.5%, respectively (p.o.).
Reference: https://doi.org/10.1021/acs.jmedchem.2c01434
M4205
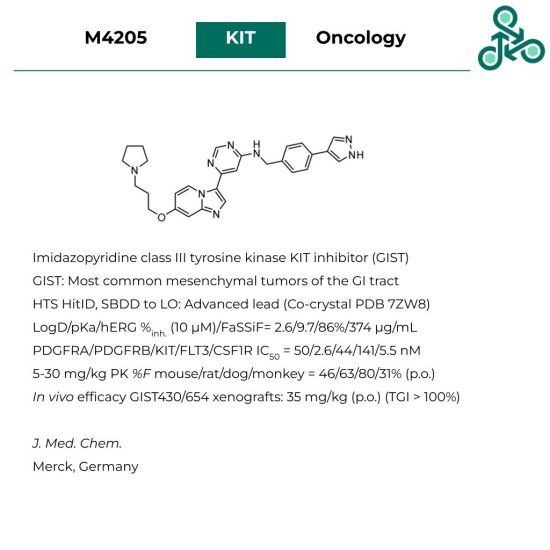
M4205: This imidazopyridine out of Merck (Germany) has been reported as an inhibitor KIT – a class III receptor tyrosine kinase, implicated in gastrointestinal stromal tumors (GIST). These neoplasms represent the most common mesenchymal tumors of the GI tract. Inhibitor development involved an initial high throughout screen towards a validated hit, after which structure-based drug design was used towards an optimized lead candidate (co-crystal reported PBD 7ZW8). In vitro, LogD/pKa/hERG %inh. (10 μM)/FaSSiF were found to be 2.6/9.7/86%/374 μg/mL. In a binding assay, against PDGFRA/PDGFRB/KIT/FLT3/CSF1R IC50 values of 50/2.6/44/141/5.5 nM were reported. In vivo oral bioavailability was observed in various animal models (mouse, rat, dog, monkey), at dose ranges between 5-30 mg/kg, with % F values of 46, 63, 80, and 31%, respectively (p.o.). In vivo efficacy was also reported in GIST430/654 xenografts, at 35 mg/kg (p.o.), with TGI > 100%.
Reference: https://doi.org/10.1021/acs.jmedchem.2c00851
RTI-122
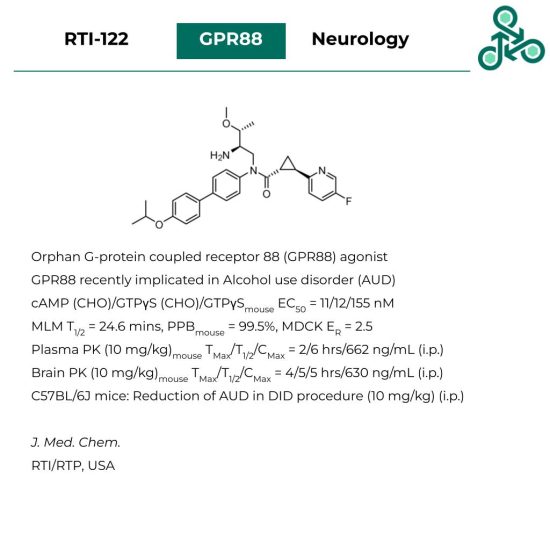
RTI-122: The Research Triangle Institute (RTI, USA) has recently reported RTI-122 as a G-protein coupled receptor 88 (GPR88) agonist. GPR88 is an orphan GPCR, implicated in Alcohol use disorder (AUD). Against cAMP (CHO), GTPγS (CHO), and GTPγS (mouse striatum), an EC50 value of 11, 12, and 155 nM was found, respectively. In vitro, in MLM, a T1/2 of 24.6 mins was observed with PPBmouse of 99.5%, and an MDCK efflux ratio of 2.5x. Pharmacokinetic analysis (10 mg/kg) in mouse plasma, found TMax/T1/2/CMax values of 2/6 hrs/662 ng/mL (i.p.). Similar trends were observed in murine brain samples. Using a drink-in-the-dark protocol (DID), in C57BL/6J mice, a reduction of alcohol drinking (i.e. AUD) was observed (10 mg/kg) (i.p.).
Reference: https://doi.org/10.1021/acs.jmedchem.2c01983
CZS-241
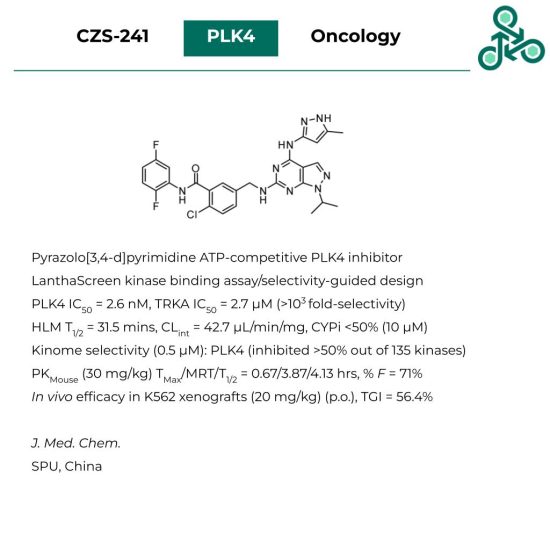
CZS-241: This pyrazolo[3,4-d]pyrimidine has been reported an ATP-competitive inhibitor of Polo-like kinase 4 (PLK4). Inhibitor design involved the use a LanthaScreen Eu kinase binding assay, as well as assessing a selectivity-guided screen against a well-known off-target (TRKA). In vitro, a PLK4 IC50 of 2.6 nM was reported, with TRKA returning an IC50 of 2.7 μM (>1000 fold-selectivity). In human liver microsomes, a half-life of 31.5 mins was found, with an intrinsic clearance of 42.7 μL/min/mg. At 10 μM, less than 50% inhibition was found against various CYP enzymes. In a kinome selectivity screen, , at 500 nM, the only target inhibited >50% (out of 135 kinases) was PLK4. In a mouse model (30 mg/kg), PK analysis found a TMax, MRT, and T1/2 value of 0.67, 3.87, and 4.13 hrs, respectively (% F = 71%). In vivo efficacy was observed in K562 xenografts (20 mg/kg) (p.o.), with a TGI of 56.4%.
Reference: https://doi.org/10.1021/acs.jmedchem.2c02124
Compound 38

Compound 38: Using molecular modelling, conformational analysis, and NMR studies towards a bioactive conformation, this molecule from AstraZeneca (UK) was recently reported as a zwitterionic selective estrogen receptor degrader (SERD) for ER+ breast cancer. Studies in estrogen receptor binding found a pIC50 of 8.8, while degradation studies found a pIC50 of 9.4, with a % DMax of 96%, and a ligand lipophilic efficiency (LLE) of 7.7. No affinity for hERG was observed (at concentrations greater than 40 μM), while pKa (BH+) values of 9.2, 7.3 were determined, along with a pKa (AH) of 2.3. The Inhibition of estradiol-induced PR expression was observed in MCF-7 cells (WT, and Y537S). In vivo efficacy was found in a CTC174 patient-derived xenograft model (50 mg/kg), with a TGI of 96%.
Reference: https://doi.org/10.1021/acs.jmedchem.2c01964
T1742

T1742: This furanylmethylene-2-pyrrolidinone has been recently reported as an autophagy-related 5 (ATG5) inhibitor. Inhibitors of ATG target critical protein-protein interactions (PPIs) between various ATGs, which have been implicated in various diseases (i.e. infections, cancer, etc.). These PPIs involve various isoforms, such as ATG12, ATG5, ATG16L1, ATG8, ATG7, and ATG10. Inhibitor design involved an initial HitID of 1500 analogs, assessed via a homogenous time-resolved fluorescence (HTRF) binding assay. Molecular dynamics and receptor-ligand docking were explored to predict a binding mode within ATG5. Against ATG5-ATG16L1, an IC50 of 1.1 μM was found, and against ATG5-TECAIR, an IC50 of 1.7 μM was observed (via HTRF binding assay).
Reference: https://doi.org/10.1021/acs.jmedchem.2c01233
AK-2292

AK-2292: The Wang group (University of Michigan, USA) has recently reported a potent, and selective PROTAC degrader of master regulator STAT5A/STAT5B using an SH2 binding ligand, and a CRBN binding lenalidomide scaffold (with utility as an anti-cancer agent against acute myeloid leukemia). Degrader design involved a structure-based approach using STAT5 SH2 domain binders via benzothiophene phosphotyrosine mimetics. In SKNO1 (18 hrs), STAT5 DC50 values of 100 nM was found, with cellular activity observed across both SKNO1, and MV4;11 cell lines. Across several AML cell lines, SKNO1, Kasumi-3, and MV4;11, DC50 (STAT5) values of 80, 50, and 180 nM were observed. A similar trend was also seen against pSTAT5Y694. In a mouse model, PK analysis revealed a T1/2 in plasma of 1.9 hrs, a clearance of 0.77 L/hrs/kg, and a volume of distribution (Vz) of 2.1 L/kg (i.p.). Finally, In vivo efficacy was observed in MV4;11 SCID mice xenografts (at 200 mg/kg) (i.p.), with a TGI of 80%.
Reference: https://doi.org/10.1021/acs.jmedchem.2c01665