In this issue of “Small Molecule Highlights” we bring you 5 new small molecules out of recent drug discovery journals. These molecules target a diverse selection of targets, including Ketohexokinase-C & A, GPR183, FLT3, PARP7, and MDM2. Enjoy!
LY3522348

LY3522348: The prevalence of high-fructose corn syrup use in the food industry and its potential implications on the incidence of diabetes and related comorbidities, such as non-alcoholic fatty liver disease (NAFLD), non-alcoholic hepatic steatosis (NASH), chronic kidney disease (CKD), and diabetic kidney disease (DKD), have inspired new ways to regulate sugar absorption and metabolism within the body. A protein family that has garnered a significant amount of interest in this space is the Ketohexokinases (KHKs). Recently, the medicinal chemistry team at Lilly optimized a highly potent hit compound (elucidation of hit was not mentioned) into an orally bioavailable and selective Ketohexokinase-C/A (KHK-C/A) inhibitor. Most of the compound’s structural features are identical to Pfizer’s clinical compound PF-06835919, likely due to low functional group tolerance within the binding pocket and tight SAR surrounding the azetidine and pyrimidine substituents. However, the scientists were able to extend a portion of the molecule using a pyrazole-linked piperazine to access a previously untapped hydrogen bonding interaction with Asp194. In biochemical assays, LY3522348 exhibited potent inhibition of human KHK-A and KHK-C (20 nM and 24 nM, respectively), and was shown to prevent the conversion of fructose to fructose-1-phosphate (F1P) in HepG2 liver cells at low nanomolar concentrations (IC50 = 41 nM). Pharmacokinetic evaluation in rat and dog revealed good oral bioavailability (83% and 87% in rat and dog, respectively) and extremely low clearance (6 and 3 mL/min/kg in rat and dog, respectively). LY3522348’s in vivo efficacy was similar to that of PF-06835919’s, with both compounds exhibiting substantial increases in serum fructose levels (10x) over control populations in an oral fructose challenge assay in lean C57/B6 mice. Furthermore, LY3522348 displayed good distribution in key target organs (kidney, liver, and heart) post study. LY3522348 has been advanced into clinical studies in humans, with Phase 1 data to be reported in due course.
Reference: https://doi.org/10.1021/acs.jmedchem.3c01410
Compound 32

Compound 32: Recently, there has been increasing evidence suggesting that G-protein-coupled receptor 183 (GPR183) plays a role in inflammatory conditions and the progression of autoimmune diseases. The authors of this paper surmised a potential application for GPR183 antagonists for the treatment of rheumatoid arthritis (RA). As most treatments for RA focus on modifying or mitigating its symptoms, a treatment that ameliorates it root cause is highly valued. Using NIBR189 (Novartis) as a chemical starting point, the medicinal chemistry team at Immunophage Biotech undertook a comprehensive SAR campaign aimed at optimizing potency, solubility, pharmacokinetic properties, and limiting hERG activity. The SAR revealed a dependence on the cinnamyl piperazine core, with key increases in potency and solubility being made by the difluoro-1,3-benzodioxole and methoxy-pyrimidine substituents, respectively. In cellular activity studies, compound 32 was shown to potently inhibit calcium mobilization (IC50 = 31 nM) and effectively blocked human U937 (IC50 = 5.5 nM) and TH17 (IC50 = 3.8 nM) cell migration in a trans-well migration assay. Pharmacokinetic profiling studies revealed low clearance in all species tested, as well as good blood exposure at low doses and >100% bioavailability in rat and dog. No toxic effects were observed in repeat-dose studies carried out in rodents and other animals (dog and monkey). In a mouse model of collagen-induced arthritis, compound 32 significantly reduced the swelling of paws and joints, decreased levels of proinflammatory cytokines and inflammatory cell infiltrate, as well as limited cartilage damage and bone erosion in affected joints. Often, the aforementioned effects were observed with doses as low as 0.1 mg/kg (p.o.). Taken together, compound 32 warrants further investigation in a clinical setting.
Reference: https://doi.org/10.1021/acs.jmedchem.3c01364
LYW713
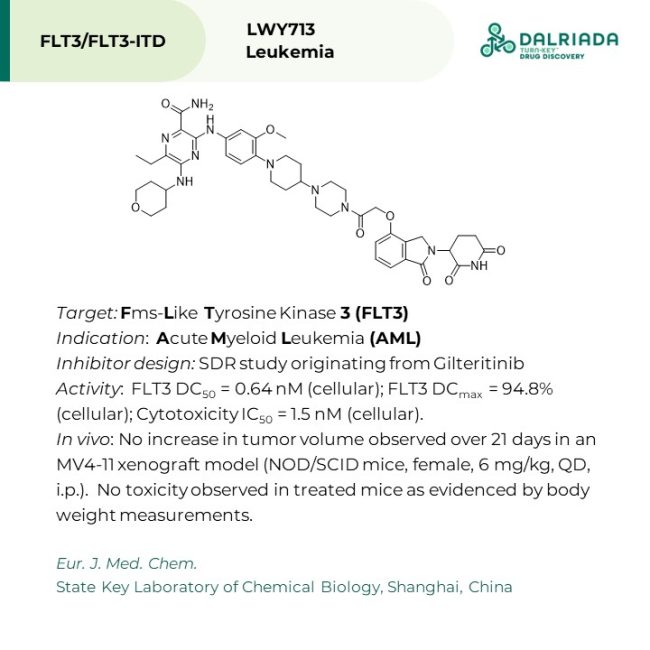
LYW713: Several first and second generation reversible FLT3 kinase inhibitors have been approved for the treatment of AML. While effective, current treatment modalities are often non-selective, as in the case of first-generation inhibitors like Sorafenib and Sunitinib, or give rise to resistance mutations which limit efficacy. As such, targeted protein degradation has been looked to as a viable option to achieve durable clinical response. Armed with a co-crystal of FLT3 with Gilteritinib (PDB:6JQR), the team at the State Key Laboratory of Chemical Biology, Shanghai, assessed an appropriate exit vector for the E3 binding ligand and performed a robust structure degradation relationship (SDR) study. The outcome, an extremely short 3 bond length linker to a lenalidomide E3 bait ligand. In a therapeutic space dominated by high molecular weight compounds, mostly due to the appendage of linker and E3 recruiting element to the ligand, atom economy is of primary importance. In MV4-11 cells, LYW718 was found to induce FLT3 degradation at sub-nanomolar concentrations (DC50 = 0.64 nM, Dmax = 94.8%), with maximum degradation efficiency being observed at 20 hours. Furthermore, the phosphorylation of downstream signalling mediators (i.e. STAT5, AKT, and Erk) was also inhibited upon LYW718 treatment. Off-target effects against AXL, ALK, and LTK (reported targets of Gilteritinib) in relevant overexpressing cell lines did not reveal credible evidence of degradation (1 and 10 nM, 2 to 20 hours). Mechanism of action via the UPS (ubiquitin-proteosome system) pathway was confirmed using the standard battery of experiments; inhibition of degradation using a proteosome inhibitor (MG-132) and loss of effect upon addition of Gilteitinib and/or thalidomide. In vivo efficacy was demonstrated in an MV4-11 xenograft mouse (NOD/SCID, female) model, with effective tumor control and slight tumor regression being observed after 21 days (6 mg/kg, QD, i.p.). The requirement for intraperitoneal injection hints at future improvements to be made to the compound to enhance its PK properties. Overall, LYW713 looks to be a promising lead compound for the targeted degradation of FLT3 and FLT3-ITD harboring AML.
Reference: https://doi.org/10.1016/j.ejmech.2023.115974
(S)-XY-05

(S)-XY-05: The discovery of small molecule STING agonists to stoke the innate immune response against various cancers has been one of the greatest applications of small molecules in the immuno-oncology space over the past decade. It has been shown that PARP7 plays a key role in cancer immunity by preventing the phosphorylation of TBK1, thereby supressing action of the cGAS-STING pathway. Towards the goal of a selective PARP7 inhibitor, the author’s drew inspiration from Ribon Therapeutics clinical candidate RBN-2397 and used a scaffold rigidification strategy to develop (S)-XY-05. Rigidification of the parent molecule was computationally guided by a combination of molecular docking and molecular dynamics simulations employing a protein structure predicted by AlphaFold. The key transformation, scaffold-hop from the original pyridazinone to indazole, conferred better selectivity, potency, and pharmacokinetic properties compared to RBN-2397. Also, the stereochemistry of the pendant methyl group was found to modulate potency, with the S-configuration being 40-fold more potent than the R-configuration. Selectivity for PARP7 was demonstrated in a panel of related proteins from the ARTD family (>70-fold selectivity observed) and further evaluated across a series of 370 kinases (no off-target inhibitory activity above 30% at 1 uM concentration). In CT-26 cells, (S)-XY-05 was found to promote phosphorylation of TBK1 and to increase the transcription of type I interferons (IFNs) and proinflammatory cytokines IFN-β and CXCL10, respectively. Pharmacokinetic evaluation of (S)-XY-05 revealed excellent oral bioavailability (F = 94.6%); a marked improvement form RBN-2397 (F = 25.7%). Dose dependant tumor growth inhibition (83% @ 25 mg/kg) was demonstrated in CT-26 xenograft mice (Balb/C) with no overt signs of toxicity. Further, analysis of resected tumors post study revealed a significant increase in CD8+T cell infiltration, implying a T-cell mediated antitumor response.
Reference: https://doi.org/10.1021/acs.jmedchem.3c01764
JN122

JN122: An example of the unexpected riches of chemistry, while developing an MDM2 inhibitor, the authors identified a spiroindoline by-product arising from an unanticipated SnAr reaction. Fortuitously, the compound bound MDM2 and MDM4 more tightly, and had better PK properties than its uncyclized predecessor. Optimization of the scaffold to enhance oral bioavailability by blocking H-bond donors and increasing steric bulk to modulate the selectivity for MDM2 over MDM4 were rational design considerations incorporated into the development of JN122. Potent antitumor activity was demonstrated across six different cancer cell lines, with p53 mediated apoptosis being confirmed by western blot in five of the six cell lines. Pharmacokinetic evaluation of the compound revealed a long serum half-life (5.35 hr for i.v. and 7.42 hr for p.o.) and a moderate oral bioavailability of 30.3%, likely due to low absorption. In vivo antitumor efficacy was evaluated in a systemic MOLM-13 xenograft mouse model, where JN122 was shown to increase the median survival time by 11-days (31 days in total) in the highest dose cohort (100 mg/kg, p.o.). Furthermore, the highest dose was well-tolerated with no apparent signs of toxicity.
Reference: https://doi.org/10.1021/acs.jmedchem.3c01815