This issue highlights 8 small molecules reported in recent medicinal chemistry literature, out of the Journal of Medicinal Chemistry, ACS Medicinal Chemistry Letters, and the European Journal of Medicinal Chemistry. Interestingly, several unique scaffolds are represented by this list of compounds, including indazoles, pyrimidines, chromanes, and urea pyrazoles. These small molecules also present several MOAs including standard inhibition, agonism, antagonism and covalency. Enjoy!

Compound 14
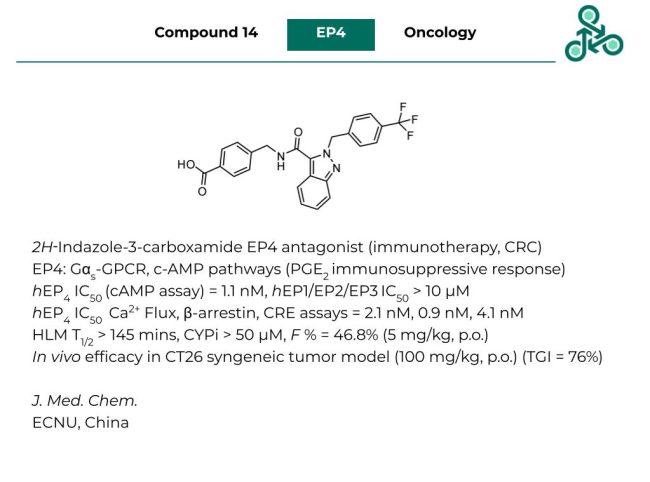
Compound 14: This 2H‑Indazole-3-carboxamide has been reported in J.Med.Chem. as an EP4 antagonist as an anticancer immunotherapy with medicinal utility against colorectal cancer (CRC). EP4 is a Gαs-coupled GPCR, with roles in cAMP pathways and PGE2 immunosuppressive responses. In a cAMP assay, a hEP4 IC50 value of 1.1 nM was reported, whilst against EP1, EP2, and EP3, an IC50 > 10 µM was found. Across various hEP4 assays (Ca2+ flux, β-arrestin, and CRE), an IC50 value of 2.1 nM, 0.9 nM, and 4.1 nM was observed. Against HLM, a T1/2 of 145 mins was found, with limited CYP inhibition (IC50 > 50 µM). At 5 mg/kg (p.o.), an oral bioavailability (F %) of 46.8% was found. In vivo efficacy was also seen in a CT26 syngeneic tumor mouse model (at 100 mg/kg, p.o.), with a TGI of 76%
Reference: https://doi.org/10.1021/acs.jmedchem.2c02058
Compound 24
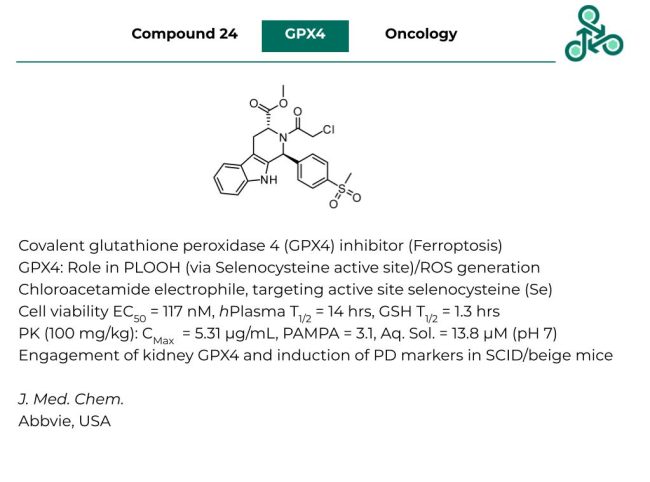
Compound 24: This small molecule by Abbvie, has been recently reported as a covalent glutathione peroxidase 4 (GPX4) inhibitor, which induces a special type of iron-dependent cell death known as Ferroptosis. GPX4 plays critical roles in lipid peroxidation, the formation of phospholipid hydroperoxides (PLOOH) along with the accumulation of reactive oxygen species (ROS). This molecule uses a reactive chloroacetamide electrophile, targeting a unique selenocysteine (Se) nucleophile within the GPX4 active site. A cell viability EC50 of 117 nM was reported, with a half-life in hPlasma of 14 hrs. Against GSH, a half-life of 1.3 hrs was found. Pharmacokinetic studies (100 mg/kg), found a CMax value of 5.31 µg/mL, a PAMPA score of 3.1, and aqueous solubility of 13.8 µM (at pH 7). Target engagement with kidney GPX4 was found, along with the induction of relevant PD markers in SCID/Beige mice models.
Reference: https://doi.org/10.1021/acs.jmedchem.2c01415
Compound 28.1
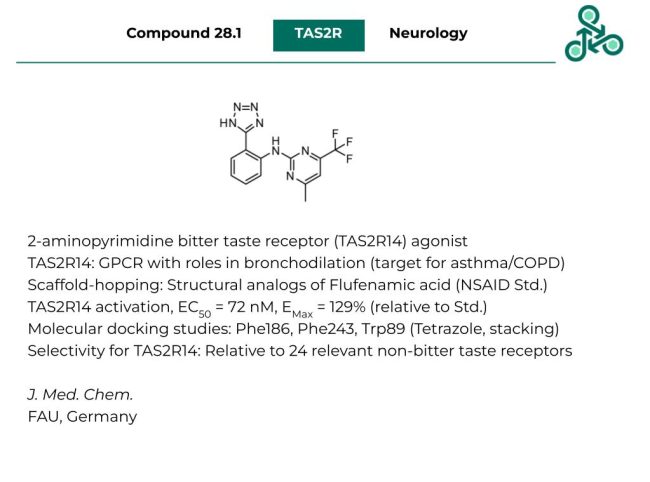
Compound 28.1: In this article, a class of 2-aminopyrimidines were found to be agonists of the bitter taste receptor (TAS2R14), out of which compound 28.1 was highlighted. TAS2R14 is a GPCR and bitter taste receptor found on the tongue, and airway passages with roles in bronchodilation. This allows it to find utility as a potential drug target for asthma and chronic obstructive pulmonary disease (COPD). Inhibitor design followed a scaffold-hopping strategy from Flufenamic acid, an NSAID clinical standard. Compound 28.1 found a TAS2R14 activation EC50 of 72 nM, with an EMax of 129% (relative to the standard). Molecular docking studies predict useful pi-pi stacking interactions between the tetrazole motif and Trp89, Phe186, and Phe243. Against a panel of 24 non-bitter taste receptors, this molecule was selective for TAS2R14.
Reference: https://doi.org/10.1021/acs.jmedchem.2c01997
CVN636
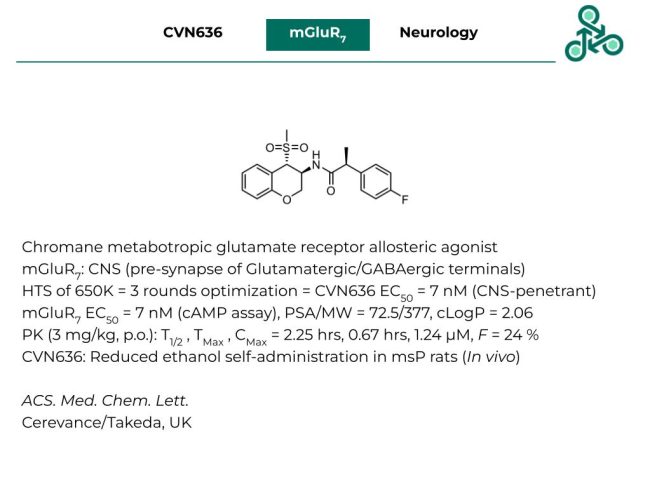
CVN636: This chromane scaffold out of Ceravance (and Takeda) has been reported as an allosteric agonist of the metabotropic glutamate receptor 7 (mGluR7). This glutamate receptor is abundantly expressed across the central nervous system, as well as pre-synaptic glutamatergic, and GABAergic terminals. Starting with a high through-put screen of 650K analogs, followed by 3 rounds of structural optimization, CVN636 was reported as a CNS-penetrant molecule, with an EC50 of 7 nM (cAMP assay). This molecule has a MW of 377 g/mol, a PSA of 72.5 Å2 and a cLogP of 2.06. In vivo PK studies (at 3 mg/kg, p.o.) revealed a half-life of 2.25 hrs, with a TMax of 0.67 hrs, and a CMax of 1.24 µM. Oral bioavailability was also observed (F % = 24 %). In an efficacy study, CVN636 reduced ethanol self-administration in msP rats (in vivo).
Compound 19
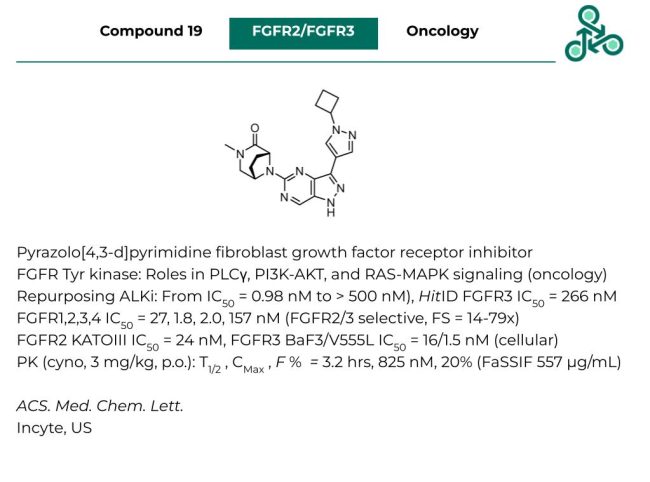
Compound 19: Incyte has recently reported this pyrazolo[4,3-d]pyrimidine as an inhibitor of the fibroblast growth factor receptor tyrosine kinase. FGFR kinases play critical roles in in PLCγ, PI3K-AKT, and RAS-MAPK signaling, with pervasive downstream roles in the incidence and progression of human cancers. Inhibitor design followed a scaffold-hopping strategy, by re-purposing a known ALK inhibitor. The initial hit identified demonstrated a FGFR3 IC50 of 266 nM. Structural optimization led to a molecule with an IC50 against FGFR1,2,3,4 of 27, 1.8, 2.0, and 157 nM (selective for FGFR2/FGFR3, 14-79x FS). In cells, against FGFR2 KATOIII, and IC50 of 24 nM was found, while against BaF3, and V555L, IC50 values of 16, and 1.5 nM were observed. Pharmacokinetics (cyno, at 3 mg/kg, p.o.) found a half-life of 3.2 hrs, with a CMax of 825 nM, and an oral bioavailability F % of 20%.
Compound 35i
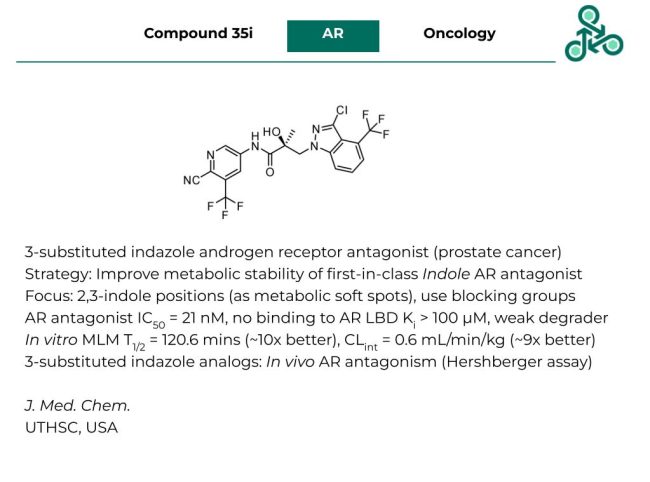
Compound 35i: This 3-substituted indazole has been reported as an androgen receptor (AR) antagonist, with medicinal use in prostate cancer. Inhibitor design strategy involved a metabolism-guided approach, improving the metabolic stability of a previously identified first-in-class indole AR antagonist via the use of blocking groups. Metabolic soft spots were identified as the 2,3 positions of the indole core-scaffold. As an AR antagonist, an IC50 of 21 nM was found, with no binding to AR LBD (Ki > 100 µM) and a weak degradation profile. In vitro stability studies in MLM revealed a half-life of 120.6 mins (10-fold improvement) and an intrinsic clearance of 0.6 mL/min/kg (9-fold improvement). Analogs of this scaffold were shown to display in vivo AR antagonism using a Hershberger assay.
Reference: https://doi.org/10.1021/acs.jmedchem.2c01858
YL5084
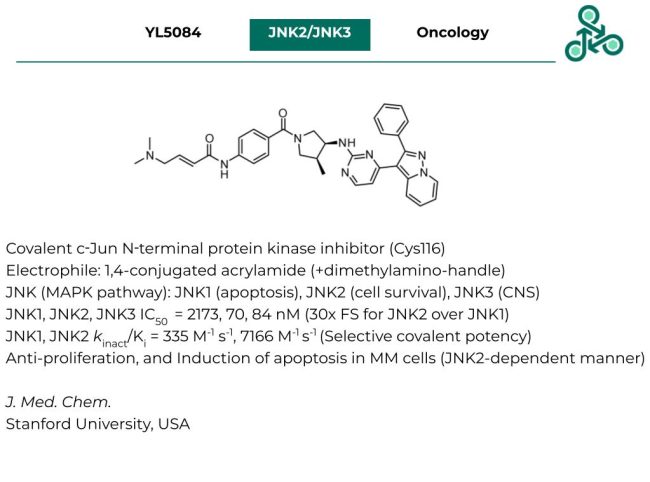
YL5084: This molecule out of the Gray lab in Stanford has been reported as a covalent inhibitor of c-Jun N-terminal protein kinase (JNK), targeting Cys116 via a canonical 1,4-conjugated acrylamide electrophile (carrying the increasingly popular dimethylamino-handle). JNK kinases, as part of the MAPK pathway involve 3 isoforms: JNK1 (implicated in apoptosis), JNK2 (implicated in cell survival), and JNK3 (roles in the central nervous system). Against JNK1, JNK2, and JNK3, IC50 values of 2173, 70, and 84 nM were reported (this represents 30-fold selectivity for JNK2, over JNK1). With regards to covalent potency against JNK1, and JNK2, kinact/Ki values of 335 and 7166 M-1s-1 were reported. Anti-proliferative activity was observed in cellulo along with the induction of apoptosis in MM cells in a JNK-2 dependent manner.
Reference: https://doi.org/10.1021/acs.jmedchem.2c01834
Compound 17b
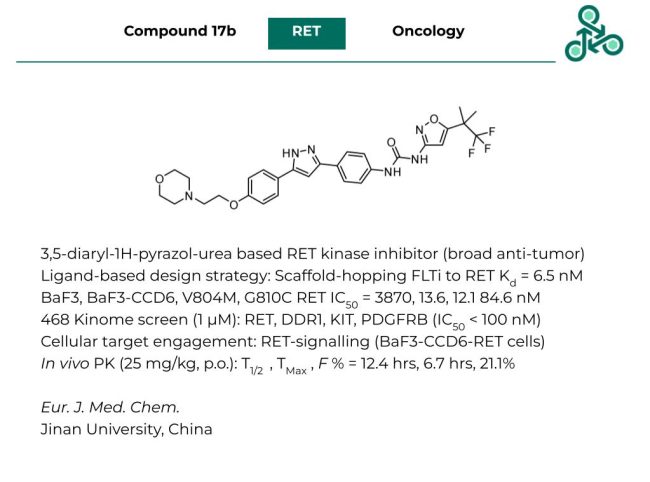
Compound 17b: This 3,5-diaryl-1H-pyrazol-urea based small molecule has been reported as a RET kinase inhibitor. Inhibitor design followed a ligand-based design strategy, using a scaffold-hoping approach from a previously identified FLT inhibitor, towards a potent RET binder, with high affinity (RET Kd = 6.5 nM). In cells, against BaF3 an IC50 of 3870 nM was observed, however against BaF3-CCD6-RET, this value went down to 13.6 nM. Against various BaF3-RET mutants (V804M, G819C), IC50 values of 12.1 and 84.6 nM were found. In a Kinome screen against a panel of 468 kinases, the only targets with IC50 < 100 nM were RET, DDR1, KIT, and PDGFRB. Cell target engagement was also confirmed, with activity against RET signalling (BaF3-CCD6-RET cells). In vivo PK analysis (at 25 mg/kg, p.o.) revealed a half-life of 12.4 hrs, a TMax of 6.7 hrs, and an oral bioavailability (F %) of 21.1%.
Reference: https://doi.org/10.1016/j.ejmech.2023.115237