In this issue of “Small Molecule Highlights” we bring you 5 new small molecules out of recent drug discovery journals. These molecules target a diverse selection of targets, including α6-nAchR, IDH2, CD38, and NLRP3. Enjoy!

CVN417

CVN417: The team at Cerevance developed this pyrazole containing small molecule to selectively modulate α6-nicotinic acetylcholine receptor (α6-nAChr) activity in the brain. This was no small feat considering the multitude of receptor/channel pentamers that can be constructed from 12 separate α and β subunits. A chemical library of 650,000 compounds was screened in a cellular calcium release assay (HTS format) to pick out hits that could be further elaborated into lead compound(s). The most promising hits contained a pyrazole core tethered via an amide linkage to a tertiary amine substituent. Multiple rounds of SAR wherein the relative placement of substituents was explored, as well as the substitution of functional groups to ones offering better brain permeability. CVN417 was characterized in microsomes and hepatocytes across 4 species, with low clearance being observed in human, rat mouse and dog. Also, low plasma protein binding (30%/20.2%/25% in human/rat/mouse, respectively) was found across all species tested. In vivo PK in rat revealed a serum half-life of 5.9 hours and a brain concentration of 1.8 μM @ 2 hours (10 mg/kg p.o.). Oral bioavailability was 11% and 43.8% in rat and dog, respectively. In a rat model of resting tremor, CVN417 was shown to reduce the duration of tremulous jaw movement dose-dependently, matching levels of efficacy found in control groups (treated with Ropinirole) at 25 mg/kg (p.o.). The authors also explored fixing sites of metabolic action on the molecule. Towards this end, the methyl groups of CVN417 were deuterated in an attempt to limit oxidative metabolism. The gamble paid off as a 4-fold improvement in stability in rat liver microsomes was observed. Unfortunately, deutero-CVN417 was not characterized in any cellular or in vivo efficacy studies, possibly hinting at a further publication where it might be the main focus.
Reference: https://doi.org/10.1021/acs.jmedchem.3c00630
ARD-1676

ARD-1676: As androgen receptor degradation is a popular therapeutic modality currently (see BWA-522 from our last post), it’s no surprise to find this latest report of an orally bioavailable PROTAC out of the Shaomeng Wang group at the University of Michigan. Building on a wealth of SAR insights from previous lead molecules ARD-2585 and ARD-2051, the Wang group synthesized a novel AR binding element containing a 5,6-spirodiamine. Interestingly, the S-methyl group on the pyrrolidine was added to occupy a small cleft within the largely hydrophobic binding domain, thereby increasing affinity. The medicinal chemists also developed a series of novel tricyclic cereblon ligands (E3 ligands). While no rationale other than novelty was given for their preparation, the thalidomide-based ligand with the fused pyrrolidine exhibited better binding affinity for cereblon than thalidomide itself. ARD-1676 was characterized in LNCaP cells against a panel of potent AR degraders, including ARV-110, where it exhibited superior degradation of AR (DC50 = 1.1 nM) and inhibition of cell growth (IC50 = 11.5 nM). Further studies using RT-qPCR confirmed suppression of AR-regulated genes in VCaP and LNCaP cells. Target selectivity was assessed in VCaP cells using standard proteomics tools where PCLAF (PCNA clamp-associated factor) and PLOD2 (procollagen-lysine 2-oxoglutarate 5-dioxygenase 2) showed up as off-target hits (>60% depletion) at 100 nM. Interestingly, these two proteins were not significantly reduced at 1 μM, suggesting a possible Hook effect. From a PK perspective, ARD-1676 exhibited good to great oral bioavailability across species, especially in non-human primates (F = 99%). A mouse VCaP xenograft model was used to assess PD. Mice receiving a single oral dose of ARD-1676 showed >90% reduction in AR protein levels by western blot analysis of tumor tissues 6- and 24-hours post treatment. Furthermore, ARD-1676 suppressed tumor growth by 85% (40 mg/kg, p.o. qd) in VCaP xenograft SCID mice without any weight loss or signs of toxicity. Taken as a whole, ARD-1676 looks to be an interesting developmental candidate in the AR+ prostate cancer space.
Reference: https://doi.org/10.1021/acs.jmedchem.3c01264
Compound 36

Compound 36: Medicinal chemists at the Jiangsu Provincial Medical Innovation Center developed compound 36 to overcome the adverse reactions observed in Enasidenib for the treatment of AML harbouring the IDH2R140Q mutation. Using a structure-based drug design (SBDD) approach, the team identified a key interaction with aspartic acid 312 that could be leveraged to obtain greater selectivity for mutant IDH2 over wildtype. Towards this end, the team incorporated an amine spacer at the 4-position of the triazine to bolster interaction with D312, and performed some light SAR centered around the substituents at the 2 and 6 positions of the triazine to develop their lead candidate. In biochemical assays, compound 36 exhibited exquisite inhibitory activity against IDH2R140Q (IC50 = 29 nM, 204 nM @ 1 hr) while sparing IDH2WT (IC50 > 100 μM @ 1 hr). In cells, the compound effectively inhibited d-2-hydroxyglutarate (D2HG) generation in TF-1 cells expressing IDH2R140Q. Pharmacokinetic analysis revealed good stability in mouse and human liver microsomes (T1/2 > 120 min) and oral bioavailability approaching 90% in BALB/c mice. Mild inhibition of hERG was observed at 30 μM (<5%). In a TF-1/IDH2R140Q xenograft mouse model, compound 36 was shown to decrease D2HG levels in tumor tissues by 62% (25 mg/kg, p.o.) with little to no impact on body weight. Given its high selectivity for IDH2R140Q and minimal inhibition of hERG, compound 36 warrants further development and evaluation.
Reference: https://doi.org/10.1021/acs.jmedchem.3c00835
Compound 1

Compound 1: The team at Immunophage Biotech developed this compound to address mitochondrial disorders affecting NAD homeostasis (mitochondrial myopathy in this case). Using a CD38 targeting compound disclosed by GSK in 2015 (compound 78c) as a starting point, the medicinal chemists used a scaffold hopping approach to find a novel pyrimidine core and optimized the attached substituents to develop compound 1. Of particular interest was the replacement of the parent thiazole moiety, a functional group thought to be necessary for binding, with imidazole, and inclusion of the methoxyethoxypyrimidine group to help with solubility and lessen cytochrome mediated metabolism. In enzymatic assays against CD38, compound 1 was found to be quite potent (IC50 = 11 and 9.8 nM in human and mouse CD38, respectively). Pharmacokinetic/ADME characterization of the compound revealed great aqueous solubility (272 μM in PBS), excellent hepatocyte stability (T1/2 > 186 min in human/rat/mouse/dog/monkey), and good oral bioavailability (F = 61/55/127/247% in mouse/rat/dog/monkey). Furthermore, there was no toxicity observed in standard safety assays (hERG, mini-Ames and 87 target safety screen). Pharmacodynamic evaluation in Pus1 KO mice (model of mitochondrial myopathy) revealed dose-dependant restoration of NAD+ levels and a salubrious effect on exercise endurance and mitochondrial biogenesis. The authors suggested potential evaluation of compound 1 in a clinical setting.
Reference: https://doi.org/10.1021/acs.jmedchem.3c00391
N14
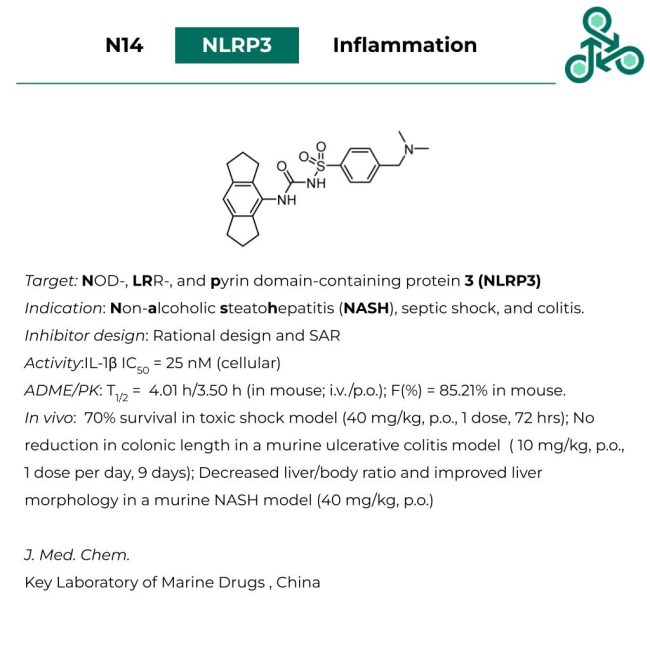
N14: Drawing inspiration from MCC950, medicinal chemists at the Key Laboratory of Marine Drugs (China) embarked on an SAR campaign to develop novel inflammasome inhibitors. Analysis of available cryoEM structures of MCC950 in complex with NLRP3 indicated that the central sulfonylurea function had to be present to maintain critical hydrogen bonding interactions within the binding pocket. The team focused on modifying the phenyl group adjacent to the sulfone to dial out liver toxicity, an issue which halted further clinical evaluation of MCC950, and to increase efficacy. Incorporation of a dimethylamino substituent at the 4-position of the phenyl group had the desired effect. N14 effectively inhibited IL-1β release in J774A.1 cells primed with LPS (IC50 = 25 nM) and didn’t induce cell death in human and mouse hepatic cells below 200 μM. Pharmacokinetic characterization revealed low clearance (38.8 mL/h/kg) and good oral bioavailability (F = 85%). Also, N14 was found to have higher and more sustained exposure than MCC950 in all tissues/organs analyzed. In in vivo mouse models of LPS-induced septic shock, ulcerative colitis, and non-alcoholic steatohepatitis (NASH), N14 alleviated symptoms in each of the aforementioned disease models. These results strongly support the further development of N14 as a therapeutic for NLRP3 inflammasome associated diseases.
Reference: https://doi.org/10.1021/acs.jmedchem.3c00894