In this issue of “Small Molecule Highlights” we bring you 5 new small molecules out of recent drug discovery journals. These molecules target a diverse selection of targets, including HBV Core Protein, HDAC1/2, BRD9, and PDE114A. Enjoy!
Lead 45

Lead 45: The team at the China Innovation Center of Roche recently disclosed this hit-to-lead compound aimed at breaking the “therapeutic ceiling” associated with current hepatitis B virus (HBV) treatment options (current options are pegylated interferons and modified nucleosides/tides). Their rationale was to target the HBV core protein, a key protein in virus replication responsible for nucleocapsid formation. High-throughput screening (HTS) of Roche’s compound collections identified a tetrahydropyrazolopyrazine (THPP) scaffold that was further optimized to increase potency, via modification of the phenyl urea substituent, and increase solubility, exemplified by the inclusion of the morpholino-lactone group. Interestingly, the team also conducted a methyl scan to explore subtle structural changes to the THPP core and its effect on virus replication. Lead 45 was shown to potently reduce HBV DNA in HepG.2.2.15 cells (EC50 = 27 nM) without any visible signs of cytotoxicity (IC50 = >100 μM in HepDE19 cells). As HBV replication occurs primarily in the liver, the medicinal chemistry team concurrently evaluated many of the compounds prepared in human and mouse liver microsomes to assess clearance. Lead 45 exhibited clearance rates of 7.1 and 39 mL/min/kg in human and mouse liver microsomes, respectively. In vivo PK in mice revealed an oral bioavailability of 48%, a high free plasma fraction (31%), and moderate liver-to-plasma exposure of 2.6-fold (p.o.). In vivo efficacy studies were conducted in a C57BL/6 mouse model mimicking HBV replication. Unfortunately, the observed efficacy (decrease in circulating HBV DNA) was modest at best and not durable. The authors mentioned that Lead 45 has been progressed to lead optimization, with the promise of highly potent inhibitors to be reported shortly.
Reference: https://doi.org/10.1021/acs.jmedchem.3c01145
Compound 17i

Compound 17i: Yet another entrant in the red-hot capsid assembly modulators (CAMs) space for the treatment of HBV, scientists at the Weifang Medical University used NVR3-778 (Novira Therapeutics) as a starting point. After an extensive SAR campaign covering five iterations of the benzamide core, the team identified a unique thioureidobenzamide (TBA) scaffold and optimized the substituents around it to prepare 17i. The compound effectively inhibited HBV DNA replication in HepAD38 cells (EC50 = 12 nM) without any acute toxicity observed within the effective dose range (CC50 = 26.6 μM; SI = 2213-fold). Furthermore, the compound potently inhibited capsid formation (IC50 = 0.88 μM) in a fluorescence based capsid assembly assay. In vitro characterization in human liver microsomes and plasma revealed half lives of 38.1 and 54 minutes, respectively. Compound 17i exhibited good oral bioavailability (F = 67.7%) and low clearance (CL = 10.30 mL/min/kg) in rat. In HBV carrier mice, 17i reduced serum levels of HBV DNA and expression of HBcAg in the liver, pointing to capsid assembly inhibition as the main mechanism of action for the compound.
Reference: https://doi.org/10.1021/acs.jmedchem.3c01022
KPZ560

KPZ560: Leveraging their HDAC small molecule discovery platform (previously used to discover selective HDAC 3 and 8 inhibitors), the team at SANKAN (Osaka University) used a combination of rational design and diversity oriented synthesis to find this selective HDAC 1/2 inhibitor. Shrewd selection of the ortho-aminoanilide as the zinc-binding substituent, and incorporation of the pendant thiophene group, hypothesized to interact with a pocket close to the catalytic site, were important design elements affording the desired selectivity. HDAC2 binding kinetics were assessed using a fluorescence-based assay wherein KPZ560 was revealed to be binding via a slow, two-step mechanism resulting in a long protein residence time (TR = 417 min). In cells, KPZ560 exhibited potent growth inhibition of MCF-7 breast cancers cells (GI50 = 43 nM) and high selectivity over normal epithelial cells (IC50 = 9.85 μM). Further washout experiments in MCF-7 cells looking at the acetylation of HDAC 1/2 target proteins confirmed the slow binding kinetics of the compound. Outside of cancer, KPZ560 was shown to increase neurite bearing cells at concentrations as low as 20 nM (15%; Neuro-2a cellular model). In vivo, mice treated with KPZ560 (1 μM, direct infusion in hippocampus) exhibited increased neurite outgrowth and density. Taken together, the author proposes KPZ560 as a novel candidate for the treatment of cancer and neurodegenerative disorders.
Reference: https://doi.org/10.1021/acs.jmedchem.3c01095
C6
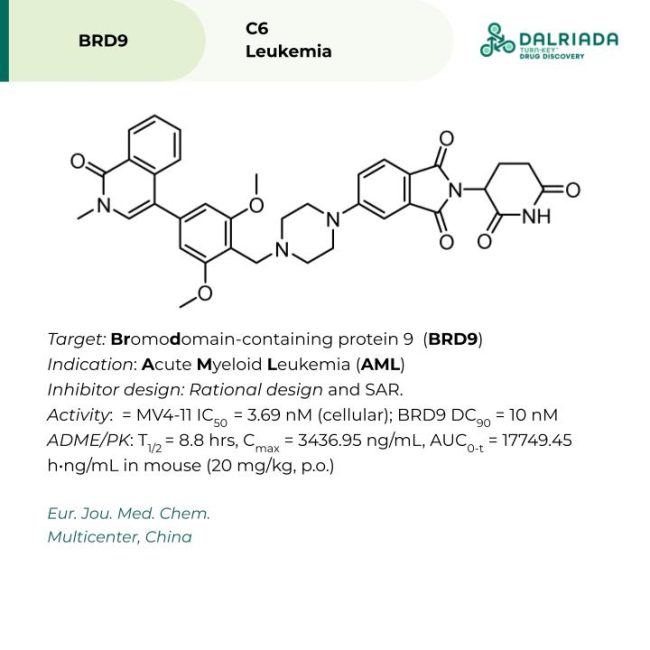
C6: This selective BRD9 PROTAC was inspired by BRD9 binder BI-7271. The medicinal chemistry team performed multiple rounds of iterative SAR to optimize the BRD9 ligand, linker, and cereblon binding substituent, with the end goal of increasing solubility/bioavailability and selectivity. Towards this end, a combination of rigid piperazine linker and C4-linked thalidomide provided the optimal balance of physicochemical properties. In MV4-11 cells, C6 was found to inhibit cell proliferation at low nanomolar concentrations (IC50 = 3.69 nM) and degrade BRD9 levels by over 90% at just 10 nM. Mechanism of action studies employing MG132 (proteosome inhibitor) and MLN4924 (ubiquitination inhibitor) confirmed C6’s dependence on the ubiquitin-proteosome system (UPS) to effect BRD9 degradation. Furthermore, selectivity for BRD9 vs BRD4 and 7 was demonstrated in MV4-11 cells via Western Blot. Pharmacokinetic characterization of the compound in mice revealed a long serum half-life (T1/2 = 8.8 hrs) and favorable plasma concentration (Cmax = 3436.95 ng/mL, AUC0-t = 17749.45 h•ng/mL) when dosed p.o. (20 mg/kg, single dose). As a whole, C6 looks like a promising lead compound for the treatment of acute myeloid leukemia (AML).
Reference: https://doi.org/10.1016/j.ejmech.2023.115872
23b

23b: The team at the Sokol Institute of Pharmaceutical Life Sciences were interested in targeting phosphodiesterase 11A4 (PDE114A) as a potential treatment modality for age related cognitive decline. Indeed, there is a wealth of literature supporting PDE114A role in memory consolidation and cognitive function. To identify a starting point, the team leveraged previously reported high throughput screening data (done in yeast) and characterized a set of HTS hit compounds in a battery of in vitro ADME assays (PBS solubility, microsomal stability, cyp inhibition, and MDCK permeability/efflux). Optimization of each of the 4 substituents attached to the pyrazolopyridine core of 23b was undertaken with the overarching goal of increasing efficacy and selectivity. In biochemical assays looking at PDE mediated hydrolysis of cAMP and cGMP, Compound 23b was found to be a potent inhibitor of PDE11A4 (IC50 = 12 nM). Furthermore, exquisite selectivity was observed for PDE11 versus PDEs 1 through 10. In a relevant hippocampal cell model (HT-22), 23b inhibited PDE114A activity at low micromolar concentrations (EC50s of 2.5 μM and 2.1 μM for cAMP and cGMP, respectively). The disparity between cellular and biochemical efficacy might indicate issues with compound solubility and/or permeability. In vitro ADME characterization of 23b revealed less than desirable aqueous solubility (42 μM in pH 7.4 PBS) and high clearance in human and mouse liver microsomes (T1/2 = 4.3 min and 3.6 min, respectively). Previous metabolism studies on the progenitor scaffold identified the diethyl amide substituent as a site of oxidative transformation. As such, the medicinal chemistry team prepared a deuteroethyl analog of 23b to circumvent this issue. Unfortunately, only a slight improvement in stability was observed for deutero-23b (compound 25 in the publication). The authors indicated their intention to further develop 23b and its congeners, with the aim of identifying an orally bioavailable candidate.
Reference: https://doi.org/10.1021/acs.jmedchem.3c01088