In this issue of Small Molecule Highlights, we bring you 7 new small molecules, fresh off the press! These compound target a diverse set of proteins (including JNK3, FXR, FABP1, P2Y6R, EGFR, MAP2K7, and more), along with utilizing interesting structural motifs: N-monomethyl carbamates, chloroacetamide electrophiles, and bicyclo[3.1.0]hexane rings. From a medicinal perspective, these molecules have been developed to target a wide-ranging list of therapeutic areas, including neurology (e.g. Alzheimer’s Disease), oncology (pediatric T-cell acute lymphoblastic leukemia), and inflammatory diseases (e.g. colitis).
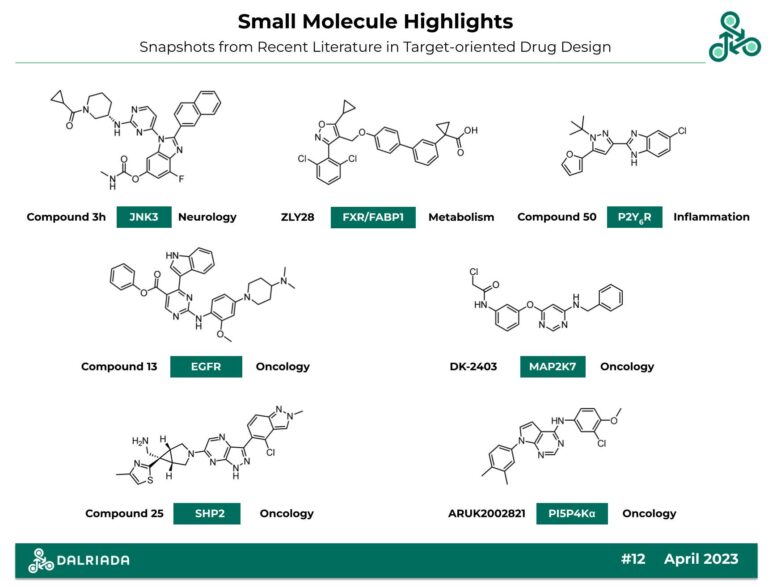
Compound 3h
Compound 3h: This N-monomethyl carbamate was recently reported as an inhibitor of c-Jun N-terminal kinase 3 (JNK3). This kinase has been implicated in Alzheimer’s Disease (AD) via interactions with APP, A β42 plaque, AMPK, Tau proteins, ultimately inducing neuronal cell death. Inhibitor design involved a pharmacodynamics (PD) focused optimization strategy. Induced-fit docking studies suggested H-bond networks with Lys68, Met149, Gln155, and a H2O molecule (PDB 4KKH). In cellular studies, neuroprotective effects were observed, along with in vivo efficacy in a 3xTg mouse model (whereby the restoration of cognitive function was observed).
Reference: https://doi.org/10.1021/acs.jmedchem.3c00393
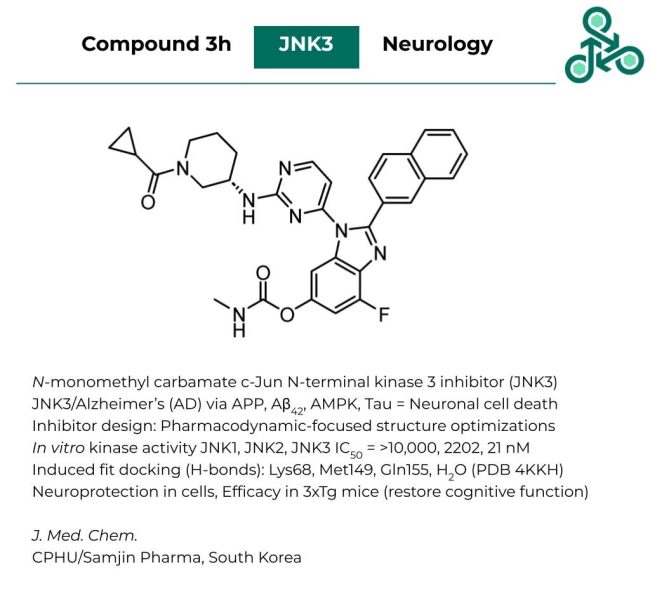
ZLY28
ZLY28: This small molecule was identified as a first-in-class dual modulator of intestinal restricted Farnesoid X Receptor (FXR) and Fatty-acid binding protein 1 (FABP1) – both of which are validated targets for non-alcoholic steatohepatitis (NASH). Inhibitor design involved a comprehensive multi-parameter approach. In a FRET assay, FXR IC50 of 143 nM was found, while cell-based studies revealed an IC50 of 491 nM. Against FABP1, this value increased to 2.7 µM. In a liver microsome stability study, the percent remaining (at 1 hour) in mouse, rat, beagle, and human microsomes was found to be 64.3, 68.6, 57.4, and 71.6%. Efficacy was demonstrated in histological models, and in vivo models of NASH/liver lipid metabolism.
Reference: https://doi.org/10.1021/acs.jmedchem.2c01918
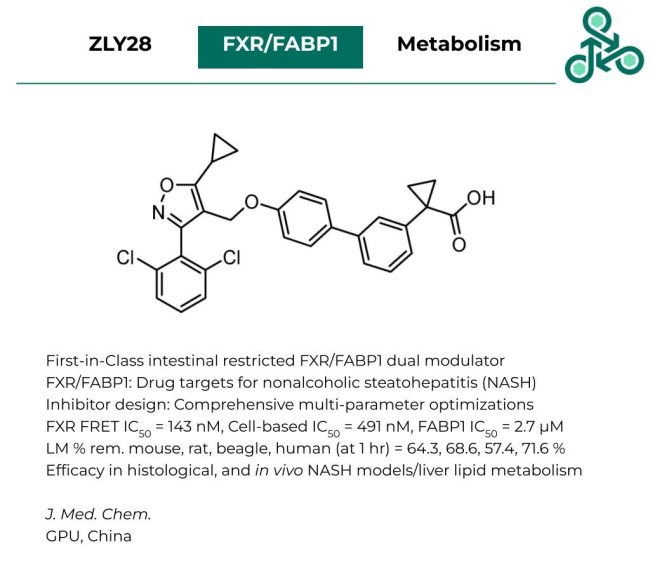
Compound 50
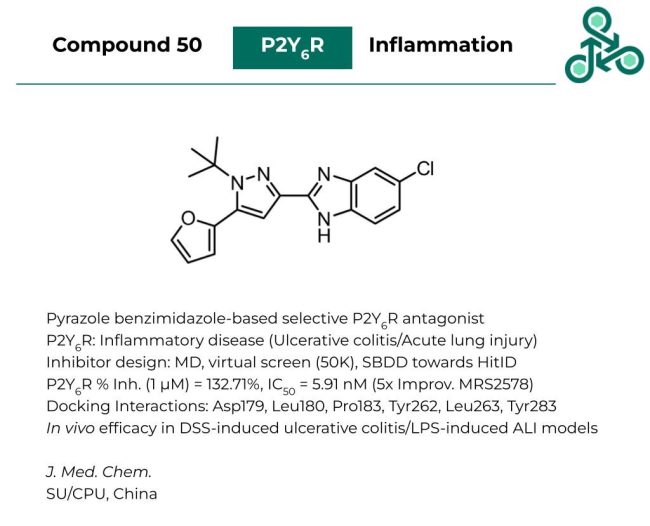
Compound 50: This pyrazole benzimidazole-based small molecule was published as a selective antagonist of P2Y6R, which has been widely implicated in several inflammatory diseases including ulcerative colitis and acute lung injury. Inhibitor design involved a multi-step process which included molecular dynamics, a virtual screen of 50,000 analogs, and a structure-based strategy towards an initial hit compound. Against P2Y6R, percent inhibition (at 1 µM) was found to be 132.71%, with an IC50 of 5.91 nM. Notably, this represented a 5-fold improvement relative to the positive control MRS2578. Molecular docking analysis revealed potential interactions with Asp179, Leu180, Pro183, Tyr262, Leu263, and Tyr283. In vivo efficacy was also demonstrated in both a DSS-induced ulcerative colitis model, as well as a LPS-induced acute lung injury model.
Reference: https://doi.org/10.1021/acs.jmedchem.3c00210
Compound 13
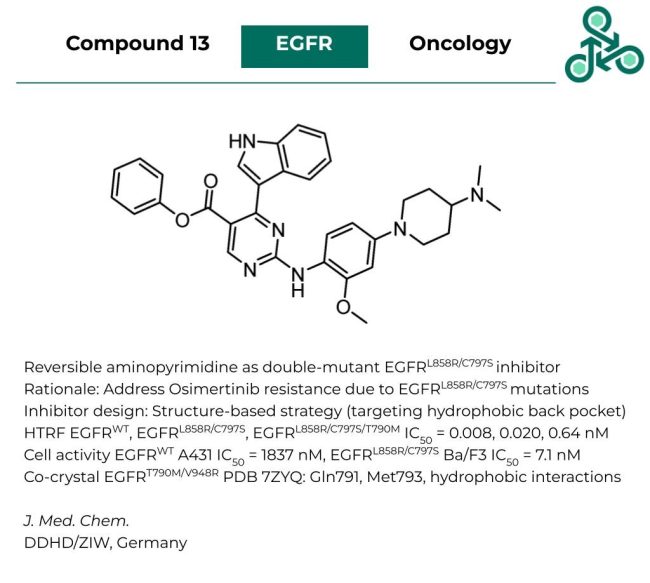
Compound 13: This reversible aminopyrimidine has been developed as a potent inhibitor of double mutant EGFRL858R/C797S as a strategy to overcome the resulting resistant to clinical standard of care Osimertinib. Inhibitor design followed a structure-based strategy targeting the hydrophobic back pocket of the kinase. In a HTRF EGFR assay, against WT, L858R/C797S, L858R/C797S/T790M, IC50 values of 0.008 nM, 0.020 nM, and 0.64 nM were reported. Cellular activity was also studied in A431 cells (EGFR-WT) with an IC50 of 1837 nM, while in EGFR-mutant dependent Ba/F3 cells, an IC50 of 7.1 nM was found. A Co-crystal structure with EGFR T790M/V948R double mutant was resolved (PDB 7ZYQ), which suggests interactions with Gln791, and Met793.
Reference: https://doi.org/10.1021/acsmedchemlett.2c00514
DK-2403
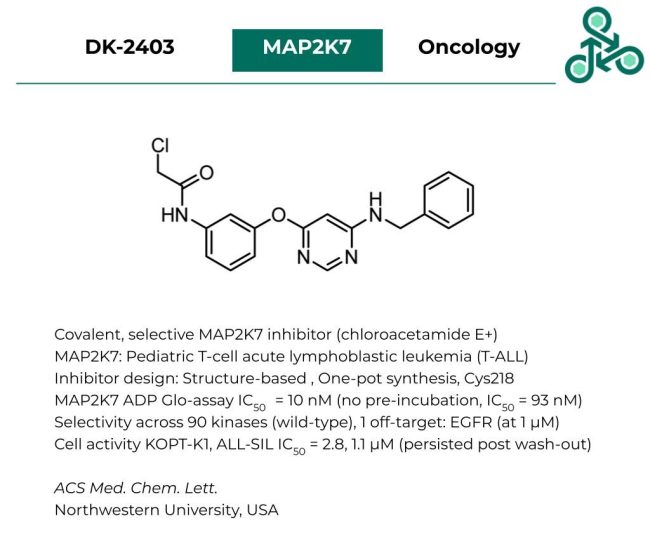
DK-2403: This small molecule was reported as a covalent and selective inhibitor of MAP2K7 using a reactive chloroacetamide electrophile. MAP2K7 has been implicated in pediatric T-cell acute lymphoblastic leukemia (T-ALL). Inhibitor design followed a structure-based strategy towards capturing Cys218. Interestingly, analog generation involved a one-pot synthesis to generate DK-2403 (and structural derivatives). In a MAP2K7 ADP-Glo assay, an IC50 of 10 nM was found, while a no pre-incubation set up resulted in an IC50 of 93 nM. Selectivity was observed against a panel of 90 kinases (wild-type), with 1 off-target, EGFR (at 1 µM). Cellular activity was also observed in KOPT-K1, and ALL-SIL cells, with IC50 values of 2.8, and 1.1 µM. This activity was also maintained in cellular-wash out studies.
Reference: https://doi.org/10.1021/acsmedchemlett.3c00029
Compound 25
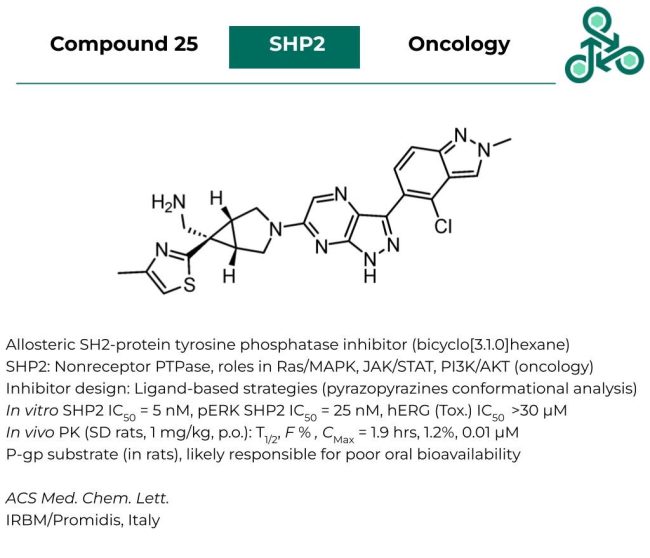
Compound 25: This structurally elaborate small molecule, with a bicyclo[3.1.0]hexane motif has been reported as an allosteric inhibitor of SH2-protein tyrosine phosphatase. SHP2 is an oncogenic non-receptor PTPase, with critical roles in several cancer-driving signaling pathways, including Ras/MAPK, JAK/STAT, and PI3K/AKT. Inhibitor design following a ligand-based strategy using several pyrazopyrazine analogs, along with conformational analysis studies. In vitro against SHP2, an IC50 of 5 nM was found, and in a pERK study, an IC50 of 25 nM was found, while limited activity was observed against hERG (IC50 > 30 µM). In vivo pharmacokinetic analysis in SD rats (at 1 mg/kg, p.o.) revealed a T1/2 of 1.9 hrs, with a weak bioavailability of 1.2 %. This is likely a result of the small molecule being a P-gp substrate (in rats).
Reference: https://doi.org/10.1021/acsmedchemlett.3c00059
ARUK2002821
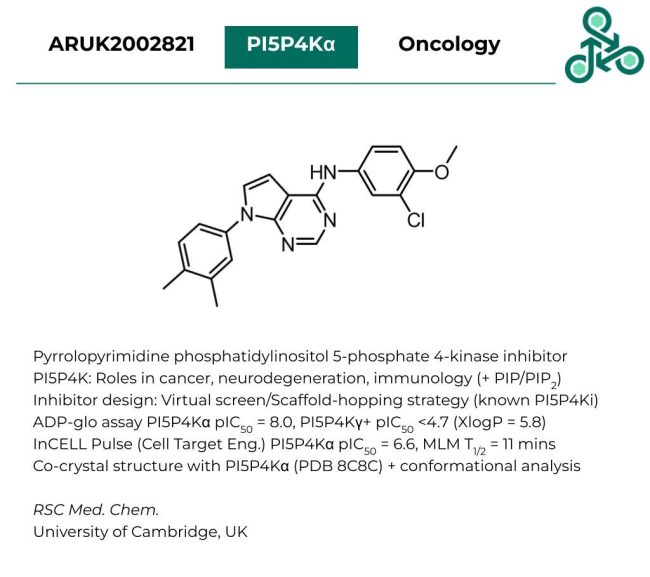
ARUK2002821: This pyrrolopyrimidine has been identified as an inhibitor of phosphatidylinositol 5-phosphate 4-kinase (PI5P4K). This kinase has diverse roles in various disease areas, including cancer, neurodegenerative disorders, and immunological ailments. Inhibitor design involved a virtual screen along with a scaffold-hopping strategy (using several well-known PI5P4K inhibitors from the literature). In a PI5P4Kα ADP-Glo assay, a pIC50 value of 8.0 was found, while against PI5P4Kγ+ an IC50 of <4.7 was reported. In an InCELL Pulse cell-target engagement study, a pIC50 against PI5P4Kα was found to be 6.6, with a T1/2 of 11 mins in mouse live microsomes. A Co-crystal was also resolved with PI5P4Kα, PDB 8C8C.
Reference: https://doi.org/10.1039/D3MD00039G