In this edition of Small Molecule Highlights, we bring an oncology-focused set of 6 ligands which have been recently identified in relevant medicinal chemistry journals. Interestingly, several of these structures also utilize unique saturated heterocycles which have been to shown to be important for target engagement. This includes a cyclopropyl group, piperazine motifs, and Oxabicycloheptanes.
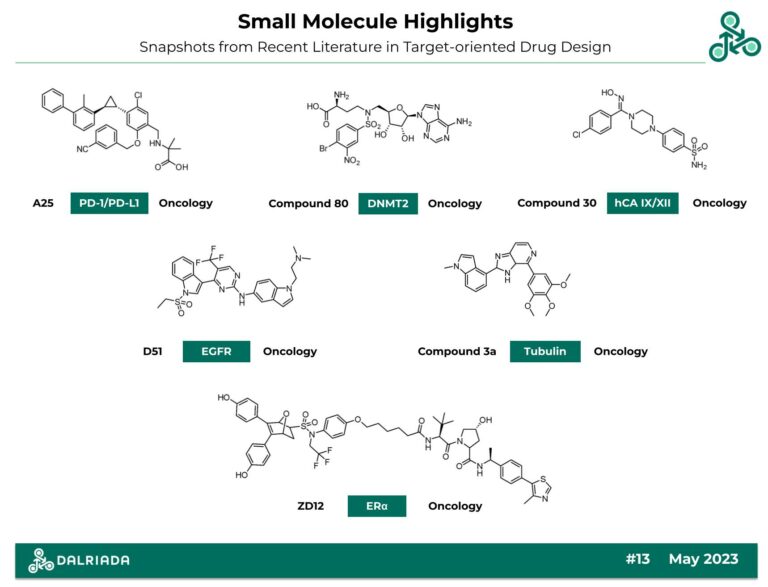
A25
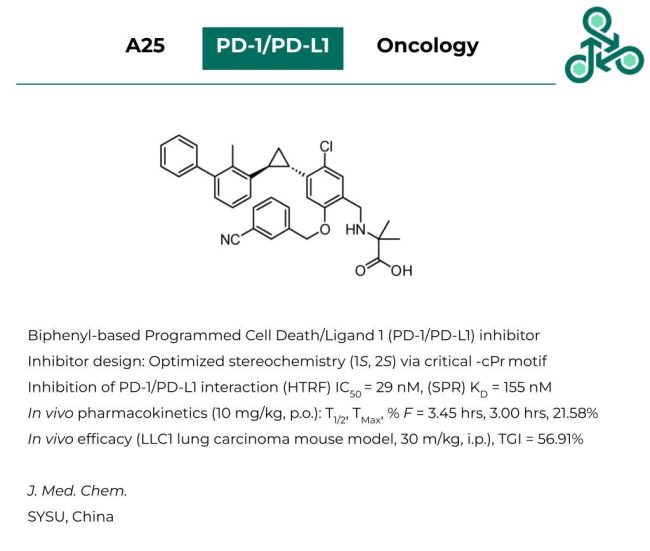
A25: This biphenyl small molecule has been recently reported as an inhibitor of programmed cell death 1 (PD-1) and programmed cell death ligand 1 (PD-L1) proteins. Structural optimization of inhibitor structure revealed the importance of a critical -cPr motif, with an optimal stereochemistry of 1S,2S. With regards to inhibiting the PD-1/PD-L1 interaction, using HTRF, an IC50 of 29 nM was reported, while in an SPR assay, a KD of 155 nM was found. In vivo pharmacokinetic analyses (at 10 mg/kg, p.o.) revealed a half-life of 3.45 hrs, a TMax of 3.00 hrs, and an oral bioavailability of 21.58%. Efficacy was also demonstrated in vivo in a LLC1 lung carcinoma mouse model, at 30 mg/kg, i.p. with a TGI value of 56.91%.
Reference: https://doi.org/10.1021/acs.jmedchem.3c00205
Compound 80
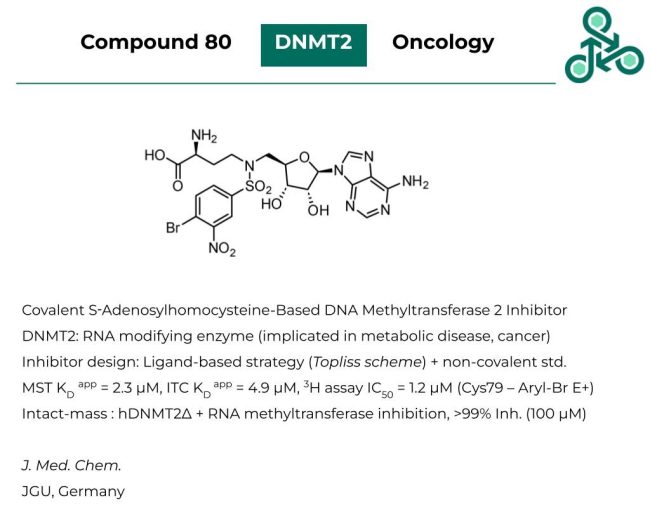
Compound 80: This molecule has been identified as a covalent inhibitor of S‑Adenosylhomocysteine-Based DNA Methyltransferase 2 (DNMT2). This RNA modifying enzyme is extensively implicated in various metabolic disorders and cancer. Inhibitor design followed a ligand-based strategy involving the use of Topliss schemes along with scaffold-analysis/hopping via a non-covalent standard. In MST, a KD value of 2.3 µM was reported, while in ITC, a KD of 4.9 µM was found, and in a 3H assay, an IC50 of 1.2 µM was found. The covalent interaction involves an electron-withdrawn aryl bromide electrophile, targeting Cys79. Intact mass analysis was validated, along with RNA methyltransferase inhibition (with >99% inhibition at 100 µM).
Reference: https://doi.org/10.1021/acsmedchemlett.3c00062
Compound 30
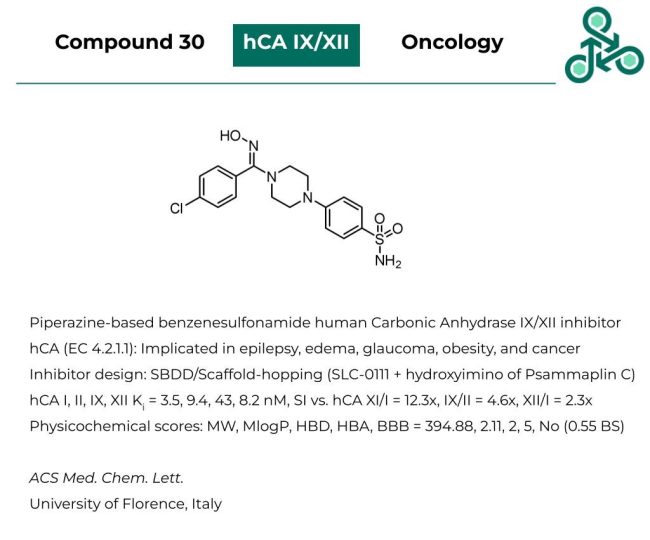
Compound 30: This piperazine-based benzene sulfonamide has been recently reported as an inhibitor of human Carbonic Anhydrase IX/XII. hCAs (EC 4.2.1.1) have been repeatedly implicated in epilepsy, edema, glaucoma, obesity, and cancer. Inhibitor design involved a structure-based approach along with several scaffold-hopping strategies (via SLC-0111 and the hydroxyimino- group seen in Psammaplin C). Against hCA I, II, IX, XII a Ki value of 3.5, 9.4, 43, and 8.2 nM was reported. In a physicochemical sense, the molecule has a MW of 394.88, with a MlogP of 2.11, HBA/HBD values of 5/2 and limited predicted blood-brain-barrier penetrance.
Reference: https://doi.org/10.1021/acsmedchemlett.3c00094
D51
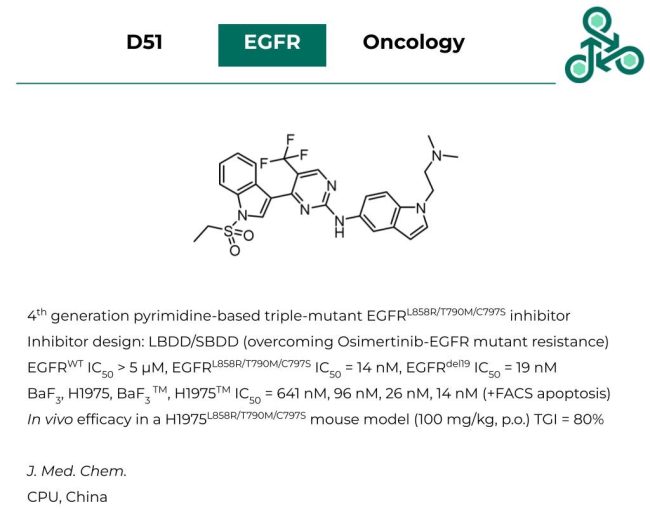
D51: This 4th generation pyrimidine-based small molecule has been identified as a reversible triple-mutant active EGFR-L858R-T790M-C797S inhibitor. Inhibitor design followed both ligand-based and structure-based strategies with a goal of overcoming the EGFR-mutant resistance observed in clinical standard Osimertinib. Against EGFR-WT, an IC50 of > 5 µM was reported, while against EGFR-L858R-T790M-C797S an IC50 of 14 nM was found. Against EGFR-del19, the IC50 value was 19 nM. In a cellular context, against BaF3, H1975, BaF3 TM, and H1975TM the IC50 values were found to be 641 nM, 96 nM, 26 nM, and 14 nM. FACS analysis also confirmed that this molecule induces cellular apoptosis in relevant cell lines. Finally, in vivo efficacy was also established in a H1975- L858R-T790M-C797S mouse model (at 100 mg/kg, p.o.) with a TGI value of 80%.
Reference: https://doi.org/10.1021/acs.jmedchem.3c00277
Compound 3a

Compound 3a: This bi-aryl indole-based small molecule has been reported as a tubulin-targeting Colchicine-Binding Site Inhibitor (CBSI). α/βTubulin play critical roles in the cytoskeleton, and are ultimately implicated in cell motility, cell growth, as well as oncogenesis and cancer metastasis. Inhibitor design involved a structure-based approached (using a co-crystal structure, PDB 7DBA), along with the use of bio-isosterism and scaffold-hopping. Against various cell lines, such as MCF-7, B16-F10, HeLa, HePG-2, IC50 values of 6 nM, 2 nM, 9 nM, and 1 nM were found (which was an improvement relative to endogenous ligand Colchicine. Several mechanistic studies were performed including X-ray crystallography (PDB 8HUH), FACS, cell-cycle arrest (HepG-2), as well as SPR (KD = 18.3 µM). In an in vivo B16-F10 melanoma model, growth was inhibited when this molecule was co-administered with PD-1/PD-L1 inhibitor NP19, at 5 mg/kg) with a TGI value of 77.85%.
Reference: https://doi.org/10.1021/acs.jmedchem.3c00011
ZD12
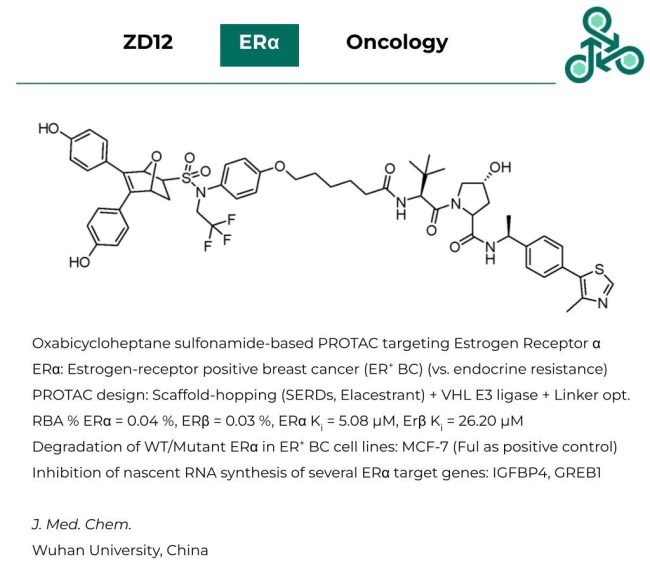
ZD12: This structurally unique Oxabicycloheptane sulfonamide-based PROTAC has been recently identified as a degrader of Estrogen Receptor α. Erα is extensively implicated in esterogen-receptor positive breast cancer (ER+ BC). PROTAC design involved a scaffold-hopping strategy, based on the success of previous SERDs and Elacestrant, along with the use of a VHL E3 ligase and structural linker optimization. In RBA, % ERα was found to be 0.04 %, while ERβ was found to be 0.03 %. As for Ki values, ERα was found to be 5.08 µM, while Erβ was found as 26.20 µM. Degradation of both the WT and mutant Erα was validated in ER+ BC cell lines (MCF-7), as well as the inhibition of nascent RNA synthesis of several ERα target genes, such as IGFBP4 and GREB1.
Reference: https://doi.org/10.1021/acs.jmedchem.2c02032