In this installment of Small Molecule Highlights, we bring you eight new small molecules, all out of the Journal of Medicinal Chemistry. These analogs have been found to act on a diverse set of cellular targets including CYP51, TRPV1, CSF1R, Skp2, BTK, and EZH2. These binding events involved standard inhibition strategies, covalent targeting, and the use of agonist effects. Several of these examples have also shown in vivo efficacy in relevant tumour-models. Enjoy!

S-F24
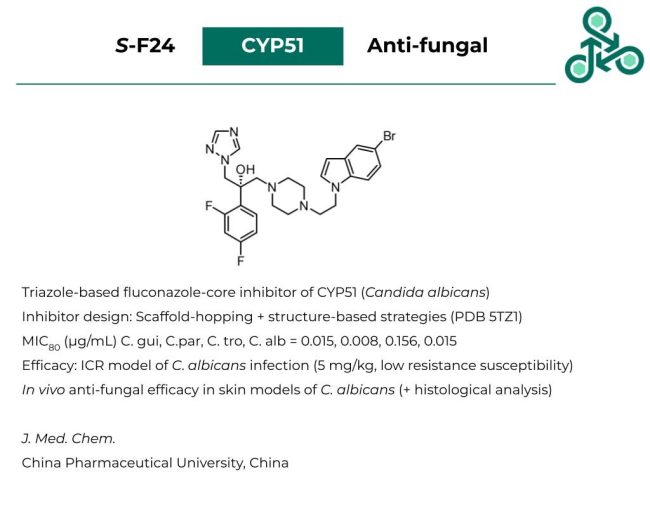
S-F24: This triazole-based small molecule with a fluconazole-core has been recently reported as an inhibitor of CYP51, implicated in targeting Candida albicans. Inhibitor design followed a scaffold-hopping and structure-based design strategy (via PDB 5TZ1). Against C. gui, C.par, C. tro, and C. alb., MIC80 values of 0.015, 0.008, 0.156, and 0.015μg/mL were reported. Efficacy was also demonstrated in an ICE mouse model of C. albicans infections (at 5 mg/kg) with low susceptibility to developing resistance (relative to other clinical standards). In vivo anti-fungal efficacy was also found in skin models of C. albicans, which was also verified via histological analysis.
Reference: https://doi.org/10.1021/acs.jmedchem.3c00266
Compound 79
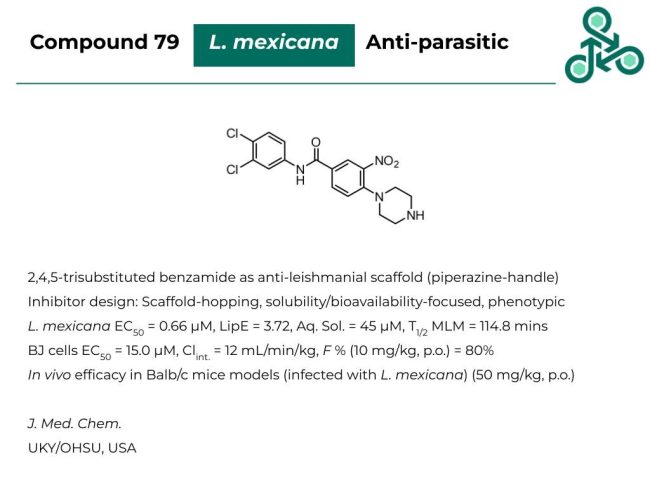
Compound 79: This 2,4,5-trisubstituted benzamide with a piperazine solubility handle has been recently reported as an anti-leishmanial scaffold. Inhibitor design was focused on improving solubility, and bioavailability, as well as based on phenotypic screening approaches. Against L. mexicana an EC50 of 0.66 μM was found, with a LipE of 3.72, an aqueous solubility of 45 μM with a half-life of 114.8 minutes in mouse liver microsomes. In BJ cells, an EC50 of 15.0 μM was reported, with an oral bioavailability (at 10 mg/kg, p.o.) of 80%. In vivo efficacy was found in Balb/c mouse models infected with L. mexicana at 50 mg/kg (via oral administration).
Reference: https://doi.org/10.1021/acs.jmedchem.3c00056
Compound 3a
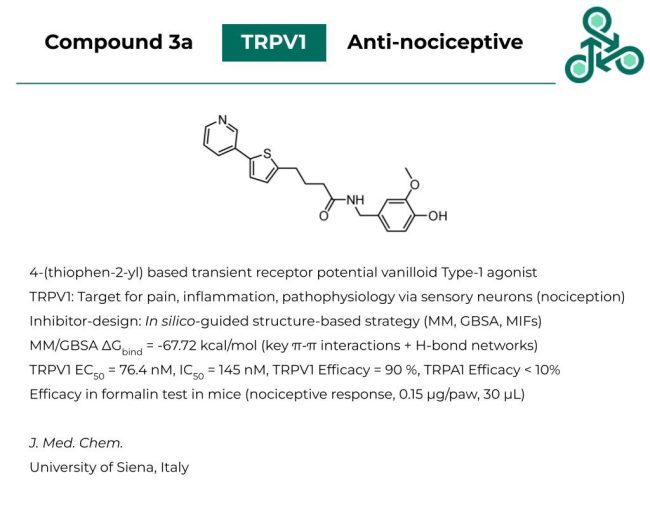
Compound 3a: A 4-(thiophen-2-yl) based molecule was published as an agonist of transient receptor potential vanilloid Type-1 (TRPV1), a validated target for pain, inflammation, and sensory neuron pathophysiology (e.g. nociception). Inhibitor design followed an in silico-guided structure-based approach via MM, GBSA, and MIFs). In MM/GBSA, a ΔGbind of -67.72 kcal/mol was found, and binding pose analysis predicted several important π-π interactions and H-bond networks. Against TRPV1, an EC50 of 76.4 nM, with an IC50 of 145 nM, and a TRPA1 efficacy of less than 10%. Efficacy in a formalin test (in mice) demonstrated a nociceptive response, at 0.15 μg/paw and 30 μL.
Reference: https://doi.org/10.1021/acs.jmedchem.3c00447
Compound 14i
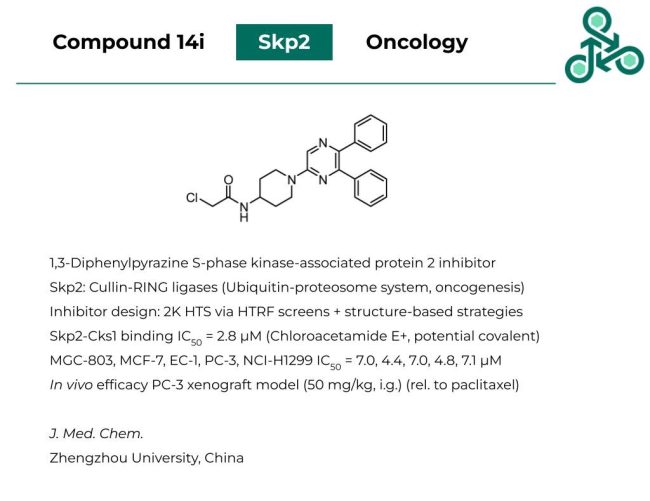
Compound 14i: This 1,3-diphenylpyrazine was found to be an inhibitor of S-phase kinase-associated protein 2 (Skp2), which is involved in Cullin-RING ligases, the Ubiquitin Proteosome System (UPS) and oncogenesis/cancer progression. Inhibitor design involved an initial 2,000 analog high-throughput screen via HTRF, followed by structure-based optimization efforts. As for Skp2-Cks1 binding, an IC50 of 2.8 μM. Interestingly, this molecule does contain a well-established covalent electrophile, with the chloroacetamide, however the authors have not commented on this potential mechanism. Against MGC-803, MCF-7, EC-1, PC-3, and NCI-H1299 cells, IC50 values of 7.0, 4.4, 7.0, 4.8, and 7.1 μM were reported. Finally, in vivo efficacy was also established in a PC-3 xenograft mouse model (at 50 mg/kg, i.g.).
Reference: https://doi.org/10.1021/acs.jmedchem.2c01675
BBDDL2059

BBDDL2059: This pyridone-based small molecule has been recently reported as a covalent and non-competitive inhibitor of enhancer of zeste homologue 2 (EZH2), involved in the catalytic subunit of PRC2 (implicated in methyl transfer via SAM to H3K27, as well as gene transcription, and the incidence/progression of cancer (upon dysregulation). Inhibitor design followed both a scaffold hopping and structure-based strategy (focused on the covalent interaction between Cys663 and the acrylamide electrophile). Against EZH2 (Y641F), an IC50 of 1.5 nM was found, while against KARPAS422 an IC50 of 64 nM was reported, with a half-life of 33.8 minutes in human liver microsomes. To validate covalency, several strategies were utilized, including IP-MS (Cys modification), Western-blot analysis (with cellular washouts), as well as CADD analysis to verify covalent vector trajectories/distances. In kinetic binding experiments, this molecule was validated as non-competitive with cofactor S-adenosylmethionine. Potency was also observed in various lymphoma cell lines, including Pfeiffer, KARPAS422, SU-DHL-10, RL, and HT, with IC50 values of less than 1 μM.
Reference: https://doi.org/10.1021/acs.jmedchem.3c00504
Compound 23
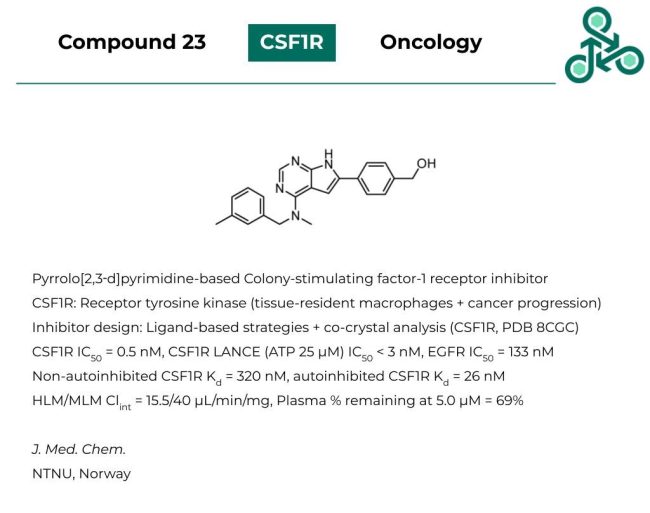
Compound 23: This pyrrolo[2,3‑d]pyrimidine has been developed as an inhibitor of Colony-stimulating factor-1 (CSF1R), a receptor tyrosine kinase implicated in tissue-resident macrophages and the incidence/progression of several human cancers. Inhibitor design followed a ligand-based strategy, before resolving a co-crystal with CSF1R (PDB 8CGC). Against CSF1R, an IC50 of 0.5 nM was found, while in a CSF1R LANCE (with ATP at 25 μM), an IC50 < 3 nM was found, and 133 nM against EGFR. In non-autoinhibited CSF1R, a Kd value of 320 nM was found, while against autoinhibited, this was reported as 26 nM.
Reference: https://doi.org/10.1021/acs.jmedchem.3c00428
SR-4133
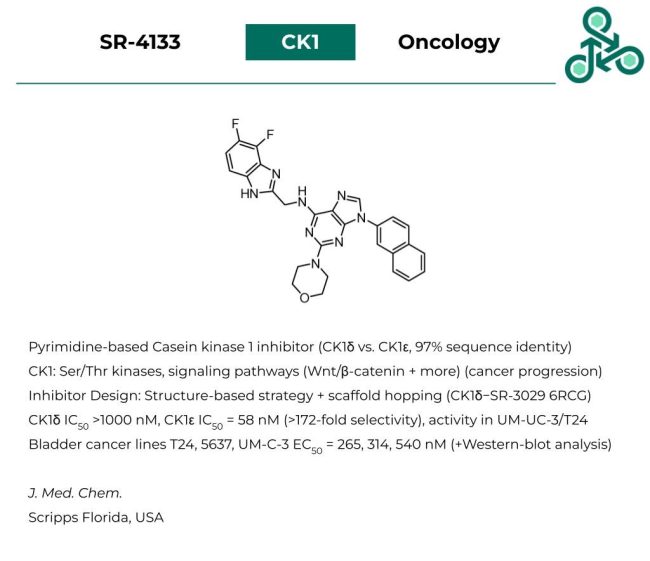
SR-4133: This pyrimidine-based analog has been reported as an inhibitor of Casein kinase 1 (CK1), which is a Ser/Thr kinase involved in several signal transduction pathways (such as Wnt/β-catenin) as well as the incidence and progression of human cancers. Inhibitor design was largely focused on a structure-based strategy, to achieve isoform-selectivity between CK1δ and CK1ε (which share roughly 97% sequence identity in the active sites). Against CK1δ, an IC50 of > 1000 nM was found, while against CK1ε, this value was found to be 58 nM (representing >172-fold selectivity for one isoform over the other. Cell activity was also found in various bladder cancer cell lines, including T24, 5637, and UM-C-3, with EC50 values of 265, 314, and 540 nM.
Reference: https://doi.org/10.1021/acs.jmedchem.2c01180
PTD10
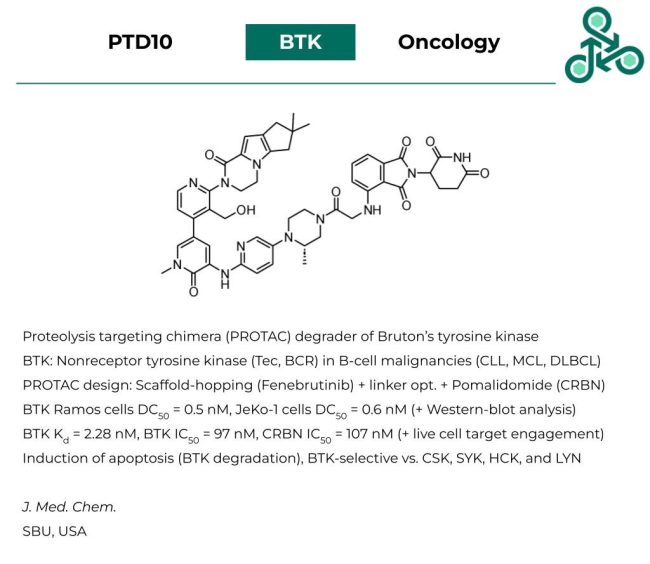
PTD10: This proteolysis targeting chimera (PROTAC) has been developed as a potent degrader of Bruton’s tyrosine kinase (BTK), which is a non-receptor tyrosine kinase (Tec/BCR) implicated in B-cell malignancies such as CLL, MCL, and DLBCL. PROTAC design involved a scaffold-hopping strategy using a known reversible BTKi Fenebrutinib, along with linker structural optimizations and the use of a pomalidomide CRNB E3 ligase. Against BTK, in Ramos cells, a DC50 of 0.5 nM was found, in JeKo-1 cells, a similar value of 0.6 nM was found. In vitro, against BTK, a Kd of 2.28 nM was reported, an IC50 of 97 nM, and a CRBN IC50 of 107 nM. The induction of cellular apoptosis was coupled with BTK degradation. In addition, this degrader was found to be selective for BTK over related proteins such as CSK, SYK, HCK, and LYN.
Reference: https://doi.org/10.1021/acs.jmedchem.3c00176