The following list of molecules represent a diverse set of structural strategies to achieve target affinity. These include dual-mode inhibition, peptide-based structures, and bi-functional binding scaffolds. Several interesting targets are highlighted below, including PHGDH, VCP/p97, MMP2, FXa, BET, HDAC, NAMPT, and IDO1. Beyond oncology, other indications are also targeted via the molecules below, these include anti-coagulants, anti-fungals, and prospective medicinal molecules against pulmonary fibrosis.

D8

D8: This small molecule was identified as a phosphoglycerate dehydrogenase (PHGDH) inhibitor, with medicinal utility as an anti-cancer therapy via the disruption of de novo serine biosynthesis (which is needed for tumor metabolism). The IC50 against PHGDH was reported as 2.80 μM, and in vitro binding assays, including microscale thermophoresis (MST), and SPR were able to define Ki values of 2.33, and 2.48 μM, respectively. Against MDA-MB-468, and MCF10A cells, IC50 values of 5.30, and 52.9 μM were observed. In ICR mice, in vivo pharmacokinetics (3 mg/kg, p.o.) revealed T1/2, Tmax, and F (%) values of 4.74 hrs, 3.33 hrs, and 82%. Finally, anti-cancer efficacy was also observed in vivo in PC-9 tumors (25 mg/kg, i.p.), with a tumor growth inhibition (TGI) percentage of 68.9%.
Reference: https://doi.org/10.1021/acs.jmedchem.2c01202
Compound 17A

Compound 17A: This pyrrolo[3,2-d]pyrimidinone was reported as a coagulation factor Xa (FXa) inhibitor, as a prospective lead compound and anti-coagulant. Against FXa in vitro an IC50 of 1.57 nM was found, whilst clinical standard Rivaroxaban (Xarelto®) observed a value of 14.38 nM. In an enzymatic activity assay (FXa), 46.7% inhibition was observed at 20 nM of inhibitor. The prothrombin time (PT %) was observed to rise in a concentration-dependent manner. Docking analysis against FXa suggested a useful hydrogen-bond between the pyrimidinone carbonyl, and the NH of Gly216 (a similar interaction found in clinical standard Apixaban). Finally, a thrombus inhibition rate of 68% was observed, at 1 mg/kg.
Reference: https://doi.org/10.1016/j.bmcl.2023.129127
Compound 19F

Compound 19F: Valosin-containing protein (VCP)/p97 represents a homohexamer, and an AAA+ ATPase, implicated in a variety of human cancers. This compound was discovered as a covalent inhibitor of p97 ATPase, targeting Cys522 in the D2 domain via an alkyne-based propiolamide electrophile. In a molecular docking analysis, a useful interaction was suggested between the NH-imidazole, and Gln473, as well as several prospective hydrophobic interactions. In an activity assay, the p97 IC50 was reported as 13.86 nM, with similar potency in U87MG glioma cells, IC50 = 19.06 nM. Using activity-based protein profiling (ABPP) pull-down assays, p97 target engagement was confirmed in cellulo.
Reference: https://doi.org/10.1016/j.ejmech.2023.115094
WZ-1831

WZ-1831: Based on an initial asparagine-ethylene diamine (AsnEDA) core scaffold, WZ-1831 was developed through a core-hopping/molecular docking strategy as an inhibitor of human constitutive proteasome chymotryptic activity (β5c). As part of the 20S proteosome, and the ubiquitin proteosome system (UPS) of degradation, this target is implicated in various cancer, specifically multiple myeloma (MM). The IC50 against β5c was found as 10 nM, while against the β5i sub-type as 7.10 μM, representing >700-fold selectivity for the desired target. Detailed SAR studies have revealed several structural motifs responsible for the observed selectivity, including the 1H-indole ketoamide, the Asn-tert-butyl structure, as well the aryl boronic acid. As for cell viability, against HepG2, MM.1S, and RPMI8226, an IC50 of 3.2 μM, 60 nM, and 2.3 μM, respectively. Finally, strong synergistic effects were observed with β5i-selective inhibitor PKS21265, which resulted in improved IC50 values <10 nM against both MM1.S/RPMI8826.
Reference: https://doi.org/10.1021/acs.jmedchem.2c00733
Compound 13C

Compound 13C: As a dual inhibitor of bromodomain and extra-terminal (BET), and histone deacetylase (HDAC) proteins, this molecule was identified for medicinal use against breast cancer, and azole-resistant C. albicans fungal infections. IC50 values against HDAC1, HDAC2, and HDAC6 were reported as 10, 152, and 9 nM, while BRD2, BRD3, and BRD4 potencies were observed as 174, 113, and 35 nM. In a cellular context against K562, HEL, HL60, and MDA-MB-231, IC50 values of 2.5, 3.9, 1.2, and 0.5 μM, respectively. Using FACS, the induction of apoptosis in MDA-MB-231 cells was evident, in a concentration-dependent manner. The blockage of C. albicans conversion was also observed in the presence of clinical standard Fluconazole (FLC).
Reference: https://doi.org/10.1021/acs.jmedchem.2c01191
Compound 10E

Compound 10E: This bi-functional molecule was reported as a dual-inhibitor of nicotinamide phosphoribosyltransferase (NAMPT), and Indoleamine 2,3-dioxygenase 1 (IDO1) as an anti-cancer strategy against NSCLC via the blockage of NAD+ synthesis. In vitro, the NAMPT IC50 = 57.7 nM, and IDO1 IC50 = 160 nM, whilst in a cellular environment in A549/R, the IC50 was observed as 5.35 μM. The inhibition of A549/R colony formation, along with the induction of ROS accumulation was observed at micromolar inhibitor concentrations. Anti-tumor activity was found in A549/R tumors (100 mg/kg, p.o.), while stronger efficacy was found in the presence of Taxol (synergistic effect).
Reference: https://doi.org/10.1021/acs.jmedchem.2c01954
TP0597850
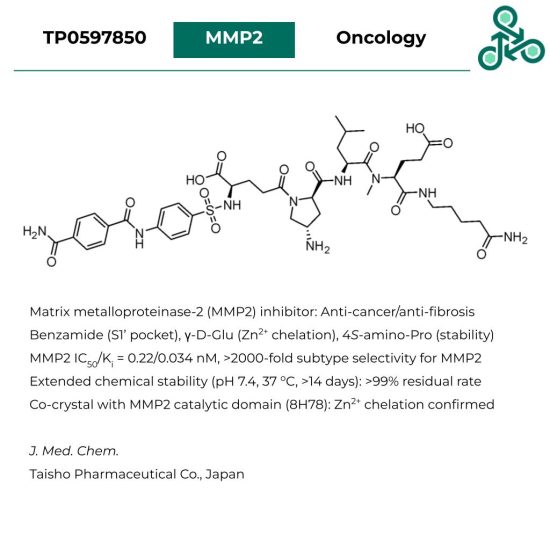
TP0597850: This peptide-based molecule is identified as a Matrix metalloproteinase-2 (MMP2) inhibitor for medicinal utility against cancer, and pulmonary fibrosis. Various structural motifs were optimized accordingly during lead identification. The phenylbenzamide is aimed to occupy the S1’ pocket, the γ-D-Glu functional group for Zn2+ coordination, while the 4S-amino-Pro group for improved stability. Against MMP2 in vitro, the IC50 was found as 0.22 nM, while the Ki value was defined as 0.034 nM, which represents >2000-fold subtype selectivity for MMP2. This peptide was also found to have extended chemical stability (pH 7.4, 37 oC) for an excess of 14 days. Finally, a co-crystal was also resolved with the MMP2 catalytic domain (8H78), which confirmed the chelation of the enzyme Zn2+.
Reference: https://doi.org/10.1021/acs.jmedchem.2c01698