The nine small molecules highlighted below feature an expansive set of target-oriented binding strategies, including dual inhibition, agonism, antagonism, targeted-protein degradation, along with the more canonical, orthosteric inhibition profile. Design efforts were largely centered around structure-based drug design (SBDD), and scaffold-hopping, focused on a diverse list of protein targets, including 1) Kinases: PI3K, LRRK2, DLK, ZLK, 2) GPCRs: GPR55, 3) Proteases: CatL/CatS, as well as 4) Chemokine receptors: CCR5.

GDC-0077

GDC-0077: This small molecule with a benzoxazepine core is an ATP-competitive phosphatidylinositol 3-kinase (PI3K) inhibitor, as well as a degrader of p110α (which is a catalytic sub-unit of PI3K). Strong selectivity (>300-fold) for PI3Kα was observed (PI3K Ki α/β/γ/δ = 0.034/99.7/18.2/12.2 nM), as well as lipophilic ligand efficiency (LLE) of 7.4. A co-crystal with p110α (PDB 8EXV) indicated H-bonds between the -CHF2– and Ser774. In HCC1954 pPRAS40, an EC50 of 19 nM was observed, along with an oral bioavailability of 58% (at 50 mg/kg, mouse). In vivo activity was found in an HCC1954 PI3KCA-mutant breast cancer (BC) xenograft model, with a TGI of 149%.
Reference: https://doi.org/10.1021/acs.jmedchem.2c01422
P7

P7: Bi-functional molecule was identified as a degrader of Son of sevenless homolog 1 (SOS1), a protein involved in RAS/MAPK signalling, which is implicated in KRAS mutant colorectal cancer (CRC). Molecular design followed a structure-based design strategy via a cereblon-SOS1 co-crystal, and the use of a lenalidomide E3 ligase. Percent degradation of SOS1 (at 1 μM, 6 hrs) in SW620 CRC cells was found to be 64% (rising up to 92% after 48 hrs). In patient-derived organoids (PDO), SOS1 degradation was maintained (1 μM, 24 hrs), representing an improvement relative to clinical standard BI3406. Finally, cytotoxicity was not observed in non-cancerous HPNE cells (up to 100 μM).
Reference: https://doi.org/10.1021/acs.jmedchem.2c01300
DN-1289

DN-1289: Dual inhibitor of both dual leucine zipper kinase (DLK), and leucine zipper-bearing kinase (LZK), which are implicated in neuronal degeneration, and amyotrophic lateral sclerosis (ALS). Biochemical assays against DLK/LZK found IC50 values of 17/40 nM, while EC50 values against DLK p-c-Jun/LZK p-c-Jun were 88/711 nM. A co-crystal of a structural analog (PDB 8DEG) found interactions with both hinge/P-loop residues. In vivo pharmacokinetic analyses revealed a T1/2 of 4.6 hrs, with an oral bioavailability of 52% (3 mg/kg, rat). PK was also determined in both mouse and cyno models. Efficacy was also observed in an acute ONC model, as well as a SOD-1 model of ALS.
Reference: https://doi.org/10.1021/acs.jmedchem.2c01056
Compound 25

Compound 25: This 1-heteroaryl-1H-indazole with a unique spiro[2.2]pentane motif, is an ATP-competitive inhibitor of leucine-rich repeat kinase 2 (LRRK2), with implications in Parkinson’s Disease (PD). A co-crystal of the LRRK2-CHK1 chimera (PDB 8E81) indicated prospective hydrophobic interactions with the spiro-nitrile functional group. In a kinase inhibition assay, an LRRK2 IC50 of 0.9 nM was found, with a Cellu IC50 of 0.3 nM. Also, a decrease of LRRK2 pS935 was found, with an EC50 of 0.18 nM. A negative AMES test was found in 5 strains, with a low projected human dose of 45 mgs.
Reference: https://doi.org/10.1021/acs.jmedchem.2c01605
Compound 24

Compound 24: As a thienopyrimidine, this molecule was identified as an inhibitor of G-protein coupler receptor 55 (GPR55), which is an orphan GPCR. Structurally, this analog originates from GPR55 antagonist ML192 (discovered via high-throughput screening). In a β-arrestin recruitment assay (in CHO cells) found an IC50 of 0.28 μM. Molecular docking analyses suggest useful hydrophobic interactions with the di-methyl motif. Limited affinity was found against both cannabinoid receptors, CB1/CB2 (at 4 μM) in a competitive binding assay. In a SRE assay, this compound was found to be a weak inverse agonist of GPR55, and in the presence of LPI, it was found to be a GPR55 antagonist.
Compound 72
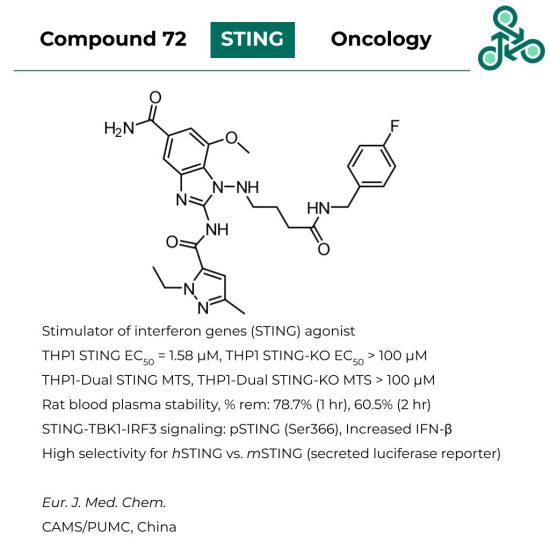
Compound 72: This small molecule was found to be an agonist of stimulator of interferon genes (STING), with implications in oncology. In THP1-dual STING, an EC50 of 1.58 μM, while against THP1-dual STING-KO the EC50 value was found to be >100 μM. MTS, in both THP1-dual STING, as well as STING-KO, the EC50 was also seen to be > 100 μM. In rat blood plasma stability study,78.7% was found (after 1 hr incubation), which 60.5% at 2 hr. STING-TBK1-IRF3 signalling was also studied, along with pSTING (Ser366), which suggested an increase if IFNβ. High selectivity was found for human STING compared to mouse STING.
Reference: https://doi.org/10.1016/j.ejmech.2022.114943
B1a

B1a: Dual inhibitor of cathepsin L (CatL), and cathepsin S (CatS) was developed as an anti-cancer strategy with anti-metastatic effects in BxPC-3, and PANC-1 cells. Inhibitor design involved a scaffold-hopping strategy (using 24 analogs), with comparative analysis against standard ASPER-29. Selectivity was observed against CatL/CatS relative to CatK (CatL/CatS/CatK IC50 = 4.10/1.79/100 μM). Evidence for anti-metastasis was found in both a wound-healing, and transwell chamber assay.
Reference: https://doi.org/10.1016/j.bmcl.2022.129087
Compound 25
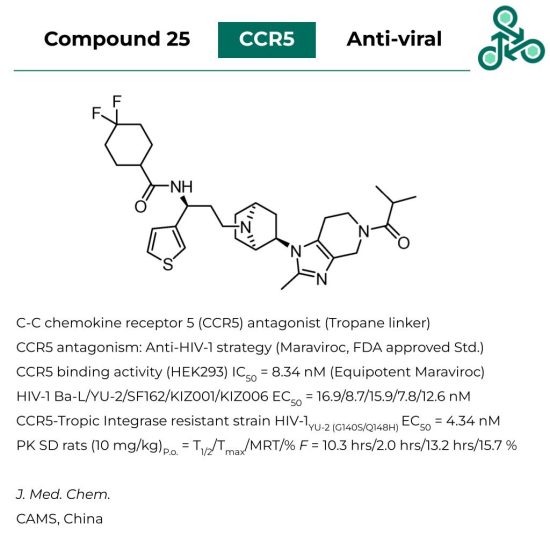
Compound 25: This small molecule, with a unique tropane linker was identified as a C-C chemokine receptor 5 (CCR5) antagonist. CCR5 antagonism has been established as a useful anti-HIV-1 strategy (as seen by the FDA approved clinical standard Maraviroc). In a HEK293 assay studying CCR5 binding activity, an IC50 of 8.34 nM (equipotent with Maraviroc). Studies against various relevant strains of HIV-1 found broad activity HIV-1 Ba-L/YU-2/SF162/KIZ001/KIZ006 EC50 = 16.9/8.7/15.9/7.8/12.6 nM. In a CCR5-Tropic integrase resistant strain HIV-1 YU-2 (G140S/Q148H), an EC50 of 4.34 nM was found. Pharmacokinetics in SD rats (10 mg/kg, orally) found a T1/2 of 10.3 hrs, a Tmax of 2.0 hrs, a MRT of 13.2 hrs, and a bioavailability (F) of 15.7 %.
Reference: https://doi.org/10.1021/acs.jmedchem.2c01383
Compound 7e

Compound 7e: This 1,2,3-triazole benzenesulfonamide was found as a human α-carbonic anhydrase (hCA) inhibitor. Structural design involved a tail-method, with hybridization with 1,3-dioxoisoindoline-5-carboxylate. Extensive studies against various CA isoforms found broad activity hCA I/II/IV/IX/XII Ki IC50 = 48.1/9.72/18.28/22.85/19.72 nM, which represented an improvement relative to the clinical standard AAZ. Molecular modelling/docking studies with hCA II (PDB 3HS4) found several interactions, including H-bonds, and pi-pi- stacking events with various residues (including Thr, Gly, Phe, and Zn2+).
Reference: https://doi.org/10.1016/j.bmc.2022.117111