In the following list of small molecule highlights, we summarize an international list of structurally novel reports out of Novartis (Switzerland), Pfizer (USA), and Merck (USA), in addition to academic institutions including CAMS/SJTU/UESTU/SU/SPU (China), OICR/PMCC (Canada), IRBM (Italy), and the University of Nottingham (UK). The molecules presented bind a varied list of drug targets, including JAK kinases, RORC2, GCS, NSD2, EZH2, BCL6, and sEH proteins (via standard or covalent mechanisms). In addition, while most entries are canonical orthosteric binders, select examples do include allosteric inhibitors, as well as partial agonists, antagonists, etc. Finally, a few examples also utilize a topical route of administration, with limited systemic potency – which introduces the concept of a soft drug.

CEE321

CEE321: This molecule was disclosed by Novartis, as a panJAKi (JAK1-3, TYK2) with therapeutic utility as an anti-inflammatory agent against atopic dermatitis (also known as Eczema). Interestingly, this small molecule observes limited systemic potency (in the blood, in vivo), while displaying good efficacy as a topical therapy – referred to as a soft drug. Enzymatic inhibitory values against JAK1, JAK2, JAK3, and TYK2 were reported as 50, 48, 59, and 50 nM, respectively. Cellular activity against IL15 (JAK1/JAK3), and IFNα (JAK1/TYK2) were found as 54, and 85 nM, respectively. Intrinsic clearance in rats via liver microsomes was found to be 204 mL/min g, while hepatocytes displayed a lower value of 94 mL/min g. Overall, a half-life of 0.5 hrs was observed. As a cream (used at 0.2% CEE321), this molecule displayed topical efficacy in the inhibition of IL4/IL13 mediated phosphorylation of STAT6, as well as MMP12 gene expression.
Reference: https://doi.org/10.1021/acs.jmedchem.2c01977
Compound 14

Compound 14: As an inverse agonist of retinoic acid receptor-related orphan receptor C2 (RORC2), this macrocycle out of Pfizer, has been reported as an anti-inflammatory topical therapy. The use of a macrocycle was based on a structure-based drug design (SBDD) strategy via co-crystal analysis (PDB 8FAV, 8FB1, 8FB2). In vitro TR-FRET based RORC2 IC50 = 9.3 nM, while a cellular IL-17 IC50 = 28 nM was found. Synthetically, ring-closure involved an Olefin metathesis step (using Grubb’s 2nd generation catalyst), after which the alkene was reduced using hydrogenation (in the presence of PtO2). Over a period of 14 hrs, topical in vitro permeation of compound 14 increases to 400 ng/cm2. In addition, 24 hrs post-application, ~90 μg/g is delivered into the epidermis, while ~25 μg/g to the dermis.
Reference: https://doi.org/10.1021/acsmedchemlett.2c00500
Compound 16

Compound 16: This bicycle pyrazole urea by Merck was reported as a low dose, orally-bioavailable, central nervous system (CNS) penetrant inhibitor of Glucosylceramide Synthase (GCS), as a medicinal agent against Parkinson’s Disease (PD). Compound 16 was discovered through a high-throughput screen (HTS), following by computational analysis, and quantitative SAR (QSAR) models. Against GCS, a biochemical IC50 of 1.8 nM was found, while 5-HT3 IC50 of 1970 nM was observed. In addition, little-to-no activity was observed against the relevant CYP enzymes (IC50 > 50 μM). In rats, in vivo pharmacokinetic analysis revealed a mean residence time (MRT) of 5.3 hrs, % F of 95%, and a VLE of 7.5. In vivo efficacy was also observed in a relevant mouse models, while ex vivo efficacy was reported in iPSC neuronal models.
Reference: https://doi.org/10.1021/acsmedchemlett.2c00441
Compound 15A

Compound 15A: The following small molecule with a structurally unique bicyclo[1.1.1]pentane linker was reported as a histone lysine methyltransferase NSD2 inhibitor. Optimizations involved the derivatization of an earlier quinoline-5,8-dione HTS hit (known as DA3003-1). Interestingly, while the MOA has not been disclosed, the molecule does contain a Cys reactive 2-butyneamide (as seen in the well-established covalent BTKi Acalabrutinib, CalquenceTM) which may suggest a prospective covalent mechanism. In vitro, NSD2 IC50 of 230 nM, while cellular activity against multiple myeloma (MM) line KMS-11 was found to display an IC50 of 270 nM. At 3.0 mg/kg (p.o.), a T1/2 of 6.71 hrs was reported. Finally, in vivo efficacy was established in KMS-11 MM tumour xenografts (in SCID mice), at 150 mg/kg (p.o.) with a TGI of 58.6%.
Reference: https://doi.org/10.1021/acs.jmedchem.2c01920
SKLB-03220

SKLB-03220: Enhancer of zeste homologue 2 (EZH2) represents the enzymatic catalytic subunit of PCR2, which plays diverse roles in histone post-translational modifications (PTMs), and the incidence/ progression of human cancers. SKLB-03220 has been reported as a covalent EZH2 inhibitor, with utility as a potential anti-cancer agent. Inhibitor design followed an SBDD strategy (via co-crystal structure analysis, PDB 5WG6). Against EZH2WT, an IC50 of 1.72 nM was reported, while against EZH2Mut IC50 values < 10 nM were found. As a covalent inhibitor, target engagement is predicted to go through a standard 1,4-conjugate addition via the acrylamide electrophile (via Cys663). Covalent target engagement was validated using koff analysis, intact-mass analysis, and cellular washout experiments. In a PA-1 xenograft tumour model (using Balb-C mice), in vivo efficacy was found, at 150 mg/kg (p.o.), with a TGI of 78.2%.
Reference: https://doi.org/10.1021/acs.jmedchem.2c01370
OICR12694

OICR12694: Through extensive structural optimization of a virtual screen weakly-binding hit compound (SPR KD = 282 μM), OICR12694 was developed as a potent inhibitor of transcriptional repressor B cell lymphoma 6 (BCL6). In vitro studies found a KD (SPR) of 5 nM (>50,000-fold improvement), and a Luc. EC50 of 89 nM, while an IC50 of 92 nM was reported in Karpas-422 cells. In mice, at 5.0 mg/kg (p.o.), in vivo PK analysis revealed a CMax of 1310 nM, a T1/2 of 1.6 hrs, and a % F of 36%. Limited CYP inhibition was observed (IC50 > 10 μM), with no observable affinity for hERG, and an AMES (-ve) test result for genotoxicity.
Reference: https://doi.org/10.1021/acsmedchemlett.2c00502
G1

G1: Through a structure-based design strategy, and scaffold-hopping, the following molecule has been developed as a Soluble epoxide hydrolase (sEH) inhibitor. This enzyme has been implicated in a variety of inflammatory diseases, including arthritis, pancreatitis, and sepsis. Inhibitor design involved a highly-extensive, and rational SAR, where various functional groups were found to be critical to the observed potency, stability, and solubility. These include the bulky memantine scaffold, the 2-fluoro group, the homo-piperazine, and the aliphatic -CH2– methylene group. Against human sEH, an IC50 of 0.05 nM (50 pM) was reported, while in murine sEH, the value was found to be 0.140 nM (140 pM). In vivo pharmacokinetic analysis in rats (50 mg/kg) (p.o.) found a TMax of 0.25 hrs, a T1/2 of 5.55 hrs, a CMax of 2.92 nM, and a % F of 20.85%. Finally, efficacy was observed (in vivo) in a CFA-induced arthritis model, an L-arginine pancreatitis models, as well as an LPS-model of sepsis.
Reference: https://doi.org/10.1021/acs.jmedchem.2c01996
Compound 10
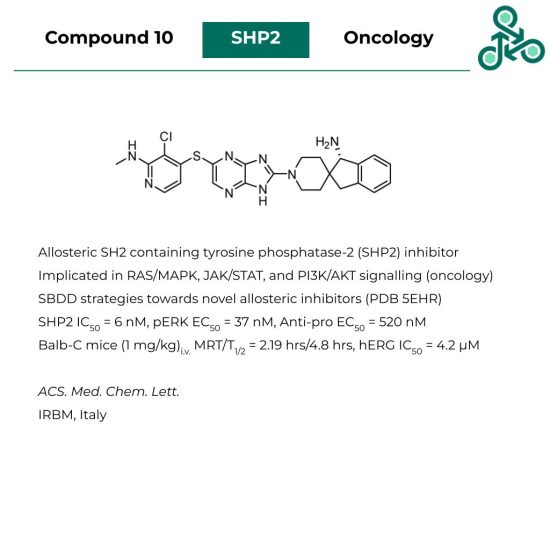
Compound 10: SH2 containing tyrosine phosphatase-2 (SHP2) is a ubiquitously expressed protein, broadly implicated in RAS/MAPK, JAK/STAT, and PI3K/AKT signal transduction (which ultimately results in a strong implication in the incidence/progression of human cancers). Compound 10 was reported as an allosteric SHP2 inhibitor, designed through SBDD strategies (PDB 5EHR). In vitro, an SHP2 IC50 of 6 nM was found, with a pERK EC50 of 37 nM, and an anti-proliferative EC50 of 520 nM. In Balb-C mice (1 mg/kg)i.v., an MRT of 2.19 hrs was reported, with a T1/2 of 4.8 hrs. Finally, against hERG, an IC50 of 4.2 μM was observed.
Reference: https://doi.org/10.1021/acsmedchemlett.2c00454
Compound 13A

Compound 13A: Using a scaffold-hopping strategy, and the derivatization of FDA approved clinical standard Cariprazine (clinical drug for schizophrenia), compound 13A was reported as a Dopamine D2/D3 receptor partial agonist/antagonist (as determined by a BRET assay). A D2R Ki value of 2.85 nM was found, while a D3R Ki value of 0.14 nM was observed (D2R/D3R = 20-fold selectivity). Against D2R GPA GαoA an EC50 of 1.70 nM was observed, while in the case of D2R mini-Gsi recruitment, an EC50 of 2.26 nM was found. In the case of D3R GPA GαoA agonism, an EC50 of 1.60 nM was reported, while for antagonism an IC50 of 4.62 nM was found. Finally, this molecule was effective in vivo in blocking cocaine self-administration in rates (1-10 mg/kg).
Reference: https://doi.org/10.1021/acs.jmedchem.2c01624