The following 7 molecules have been recently reported as molecular binders of various interesting medicinal targets, including epigenetic hydrolases (e.g. HDACs), oncology-implicated kinases (e.g. BTK), anti-viral targets (e.g. HBV), anti-psychotic GPCRs (e.g. GPR52), as well as novel targets, such as RIPK1, with prospective utility against respiratory illnesses such as acute respiratory distress syndrome (ARDS). The following articles can be found in J.Med.Chem., ACS. Med. Chem. Lett., and Eur. J. Med. Chem.

Compound 19h
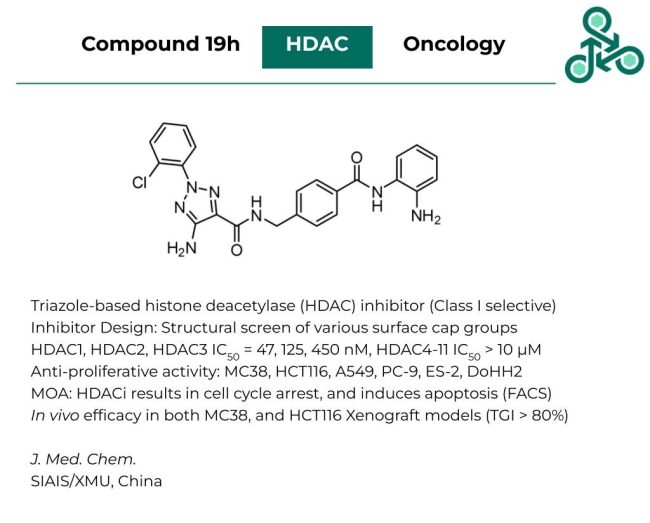
Compound 19h: The following triazole-based small molecule was reported as a histone deacetylase (HDAC) inhibitor, selective for class I isoforms: HDAC1, HDAC2, HDAC3. Inhibitor design involved a structurally focused interrogation of various surface cap groups. In biochemical assays, this compound found an IC50 of 47 nM, 125 nM, and 450 nM against HDAC1, HDAC2, and HDAC3, respectively. While an IC50 of > 10 µM was reported for the remaining deacetylase isoforms (HDAC4-HDAC11). Broad anti-proliferative activity was found across various cell lines, including MC38, HCT116, A549, PC-9, ES-2, and DoHH2. Mechanistic studies revealed this molecule results in cell cycle arrest, and FACS analysis demonstrated the induction of apoptosis. In vivo efficacy was observed in both MC38 and HCT116 xenograft models with TGI values of >80% in both cases.
Reference: https://doi.org/10.1021/acs.jmedchem.2c01985
BGB-8035
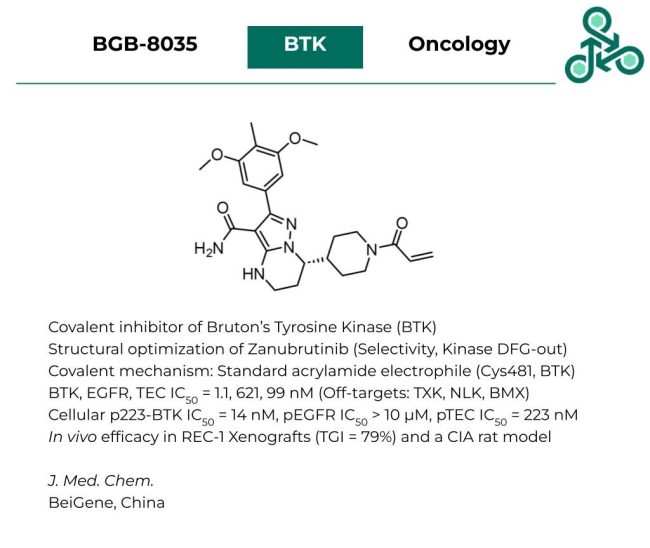
BGB-8035: This molecule was recently reported by BeiGene as a covalent inhibitor of Bruton’s Tyrosine Kinase (BTK). Inhibitor design was focused on several improvements on the well established covalent BTK inhibitor Zanubrutinib (Brukinsa®), with respect to selectivity, as well as targeting the kinase DFG-out conformation. The MOA follows a standard covalent 1,4-conjugate addition between the canonical acrylamide electrophile, and BTK Cys481. In vitro, an IC50 of 1.1 nM was found against BTK, with values of 621 nM, and 99 nM against EGFR and TEC proteins. In a kinome screen, common off-targets included TXK, NLK, and BMX. In a cellular environment, against pBTK (p223), an IC50 of 14 nM was reported, while pTEC returned a value of 223 nM, and pEGFR found an IC50 of > 10 µM. In vivo efficacy was also demonstrated in both a REC-1 xenograft model (TGI = 79%), as well as a Collagen-induced arthritis (CIA) rat model.
Reference: https://doi.org/10.1021/acs.jmedchem.2c01938
RG7907

RG7907: This structurally elaborate small molecule was recently reported by CICoR (Roche), as a Hepatitis B Virus (HBV) core protein allosteric modulator, with utility as an anti-viral therapeutic. The structural core involves a hetero-aryldihydropyrimidine, which stems from previous known HBV ligands, such as HAP_R01. In HegP.2.2.15 cells, against HBV DNA, an EC50 of 6 nM was found, while in HepDE19 cells, limited cytotoxicity was observed with a CC50 of > 100 µM. A co-crystal structure was determined against Cp149 Y132A (PDB 8I71), which highlights interactions with Trp125, and a hydrogen-bond network with a nearby H2O molecule. Extensive in vivo pharmacokinetic characterization was performed, in many models, including rat, monkey and minipig (with oral bioavailability observed across all 3 cohorts). In vivo efficacy was also observed in an adeno-associated virus-HBV mouse model (at 20 mg/kg, q.d.).
Reference: https://doi.org/10.1021/acs.jmedchem.3c00173
HTL0041178
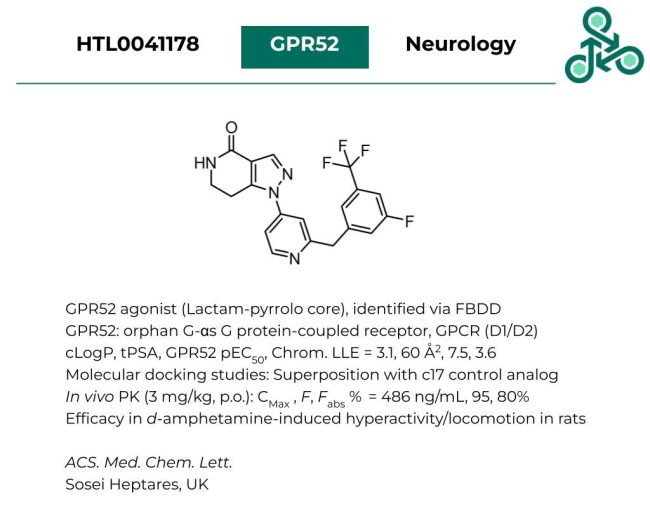
HTL0041178: The following molecule with a lactam-pyrrolo-core out of Sosei Heptares has been reported as a GPR52 agonist, identified via rational fragment-based drug discovery (FBDD). GPR42 is an orphan G-αs G protein-coupled receptor (GPCR) with critical roles in both D1, D2 signaling. Calculated physicochemical properties returned values of cLogP = 3.1, tPSA = 60 Å2 , GPR52 pEC50 = 7.5 and Chrom. LLE = 3.6. Molecular docking studies demonstrated strong superposition with control compound c17. In vivo PK analysis (3 mg/kg, p.o.) revealed a CMax of 486 ng/mL, and an oral bioavailability (F %) of 95%. Efficacy was also established in a d-amphetamine-induced hyperactivity/locomotion model in rats.
Compound 11
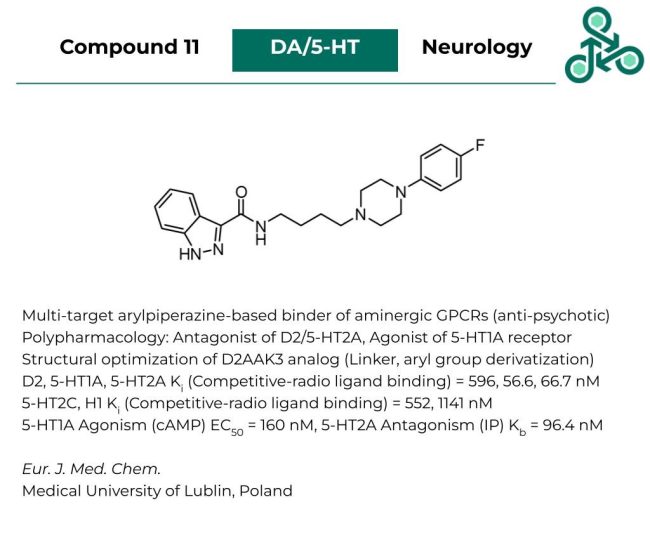
Compound 11: This arylpiperazine-based small molecule has been reported as a multi-target binder of aminergic GPCRs, with prospective utility as an anti-psychotic therapy. This polypharmacology involves antagonism against D2/5-HT2A, and agonism against the 5-HT1A receptor. Using a competitive radio-ligand binding assay, Ki values of 596 nM, 56.6 nM, and 66.7 nM were reported against D2, 5-HT1A, and 5-HT2A, respectively. Off-targets such as 5-HT2C, and H1 returned Ki values of 552 nM, and 1141 nM. Agonism against 5-HT1a (cAMP signalling) observed an EC50 of 160 nM, while 5-HT2A antagonism (IP) found a Kb of 96.4 nM.
Reference: https://doi.org/10.1016/j.ejmech.2023.115285
Compound C16

Compound C16: This structurally complex α-aminoketone 4-ring system has been reported as a dual inhibitor of p53-MDM2, and MDMX. The p53-MDM2/MDMX interactions importantly regulate the transcriptional response of p53 (also known as guardian of the genome). Through rational structural optimizations, a Ki value of 0.23 µM was reported against MDM2, and 2.54 µM against MDMX. As for anti-proliferative activity, an IC50 of 0.68 µM was found in HCT116 cells, and 0.54 µM against SH-SY5Y cells. Extensive MM/GBSA calculations were performed to study/identify binding hot-spot residues of MDM2/MDMX. In addition, the inhibition of HCT116 migration and invasion was observed, along with the re-activation of p53 activity.
Reference: https://doi.org/10.1016/j.ejmech.2023.115282
SZM-1209
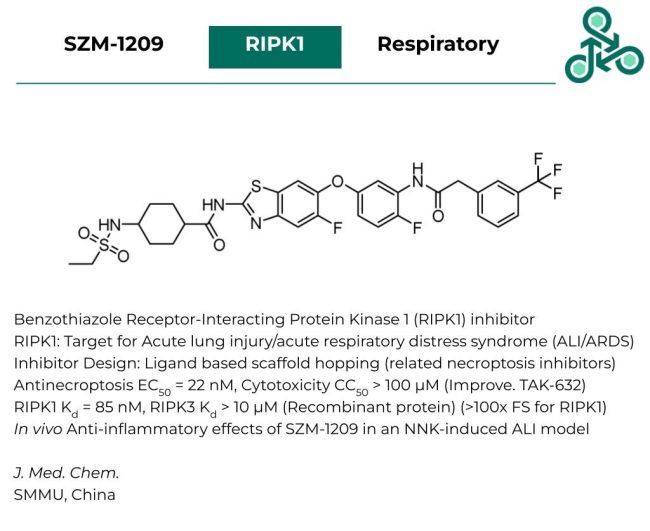
SZM-1209: This benzothiazole molecule has been recently revealed as an inhibitor of Receptor-Interacting Protein Kinase 1 (RIPK1). Interestingly, RIPK1 has been validated as one of the few targets for Acute lung injury/acute respiratory distress syndrome (ALI/ARDS). Inhibitor design utilized a ligand-based scaffold hopping strategy, based on previously established necroptosis inhibitors. Anti-necroptosis, an EC50 of 22 nM was found, while with respect to cytotoxicity, a CC50 of > 100 µM was reported (representing an improvement relative to standard analog TAK-632). Against RIPK1, a Kd value of 85 nM was found, while against RIPK3, a Kd of > 10 µM was observed. This signifies a >100x fold-selectivity for RIPK1. In vivo anti-inflammatory effects were also observed in an NNK-induced ALI model.
Reference: https://doi.org/10.1021/acs.jmedchem.3c00197