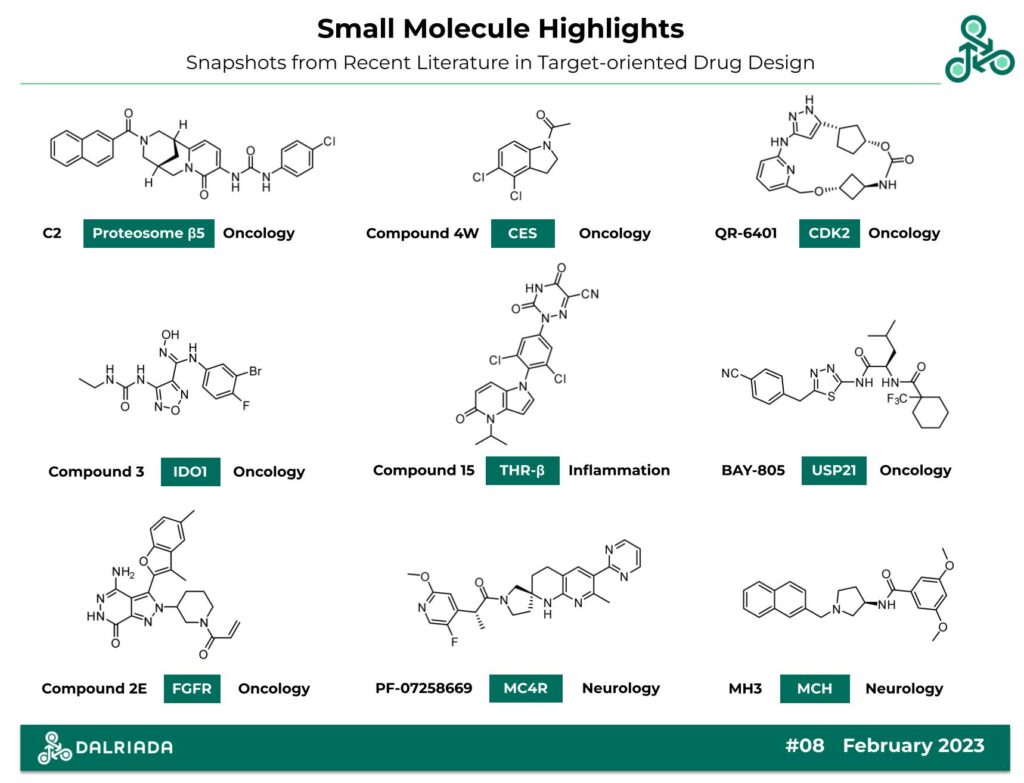
The following list of 9 molecules, out of various drug discovery journals, have been primarily identified through the use of structure-based drug design strategies (SBDD), including X-ray co-crystal analysis, molecular modelling, receptor-ligand docking, and large-scales virtual screens. Altogether, these compounds have been designed to bind a diverse set of targets, including proteosome β5, CES Notum, CDK2, IDO1, THR- β, MC4R, MCH, USP21, and FGFR. Interestingly, several of these molecules have been designed to modulate neurological circuits of appetite, weight loss, and food intake (hyperphagia, anorexia, and cachexia).
C2

C2: This naphthyl-azo-tricyclic-urea-based small molecule was recently reported as a proteosome β5 inhibitor. Through targeting the Ubiquitin-proteosome pathway (UPP), C2 is being developed for therapeutic utility in oncology, neurology, and autoimmune disease. Identified through a virtual screen of a natural product ZINC library (N = 5000) and a structure-based design strategy, a β5 IC50 of 3.43 μM was reported, with a Ki of 1.15 μM (in a purified enzymatic assay). Molecular docking predicted key intermolecular stacking interactions at the β5c site with Tyr169, and the β5i site with Phe124, and Tyr130.
Reference: https://doi.org/10.1039/D2MD00404F
Compound 4W

Compound 4W: A collaboration between the University College London (UCL), and Oxford University in the UK has recently identified this N-acyl-indoline as carboxylesterase Notum inhibitor. CES Notum has been implicated in Wnt signalling, with therapeutic utility in oncology. This small molecule was discovered through a structure-based strategy from a covalent hit compound (Methyl ester electrophile), and a co-crystal of a structural analog (PDB 8BT8). This analysis identified a stacking interaction with Phe268, as well as a bridging H2O-mediated H-bond. Against Notum-OPTS, an IC50 of 4.9 nM was reported, and in a cellular TCF-LEF assay, an EC50 of 22 nM was found. This compound has a LogD7.4 of 3.1, PBS solubility of 0.4 μg/mL and a plasma stability T1/2 of > 4 hrs.
Reference: https://doi.org/10.1016/j.ejmech.2023.115132
QR-6401

QR-6401: This macrocycle was recently reported a potent and selective inhibitor of cyclin-dependent kinase 2 (CKD2). The authors cited prospective therapeutic utility in CDK2-dependent cancers, and combating CCNE1 drug resistance. The design of this molecule was accelerated from hit-to-lead through the use of computational macrocycle generation models, and structure-based drug design. Against CDK2/E1, an IC50 of 0.37 nM was found, while against CDK1/A2, an IC50 of 22 nM was found (59x selective). In a cellular OVCAR3 viability study, an IC50 of 34 nM was found, and against human liver microsomes (HLM), a T1/2 of 150 mins was established. In OVCAR3 xenografts, at 50 mg/kg (BID) using oral gavage, a TGI of 78% was found.
Compound 3
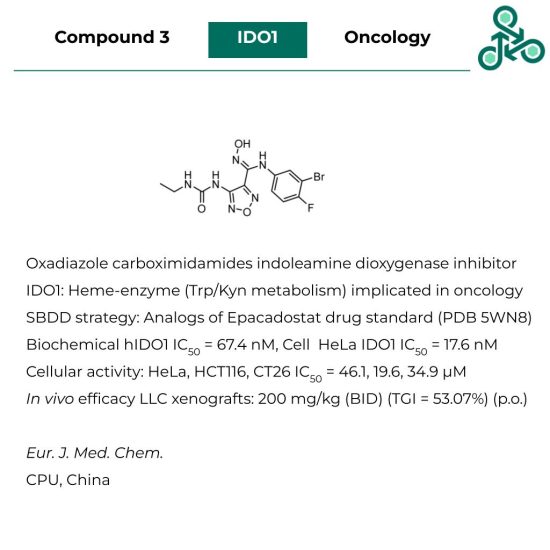
Compound 3: This Oxadiazole carboximidamide was reported as an indoleamine dioxygenase 1 (IDO1) inhibitor. IDO1 is a heme-containing enzyme, involved in Trp/Kyn metabolism and implicated in oncology. This small molecule was discovered via a structure-based strategy, through structural analogs of FDA-approved clinical standard Epacadostat (PDB 5WN8). In a biochemical assay against hIDO1, an IC50 of 67.4 nM was found, and in a cellular HeLa IDO1 study, an IC50 of 17.6 nM was reported. Weak cellular activity was observed in various cell lines: HeLa, HCT116, and CT26, with IC50 values of 46.1, 19.6 and 34.9 μM. In vivo efficacy was found in LLC xenografts at 200 mg/kg (BID) (p.o.) with a TGI of 53.07%.
Reference: https://doi.org/10.1016/j.ejmech.2023.115217
Compound 15
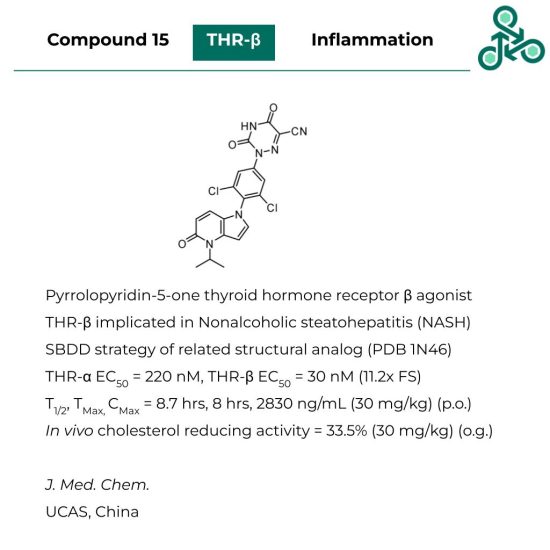
Compound 15: This pyrrolopyridin-5-one has been reported as an agonist of thyroid hormone receptor β, which is implicated in nonalcoholic steatohepatitis (NASH). Using a structure-based structure of a related structural analog (PDB 1N46), an EC50 against THR-α was found to be 220 nM, while the desired target THR- β was 30 nM (11.2x selectivity window). At 30 mg/kg (p.o.), a T1/2 of 8.7 hrs was found, with a TMax of 8 hrs and a CMax of 2820 ng/mL. In vivo, cholesterol reducing activity was observed, at 33.5% with a dose of 30 mg/kg (oral gavage).
Reference: https://doi.org/10.1021/acs.jmedchem.2c01669
BAY-805

BAY-805: Bayer recently reported this 1,3,4-thiadiazol-based small molecule as a ubiquitin-specific protease inhibitor targeting USP21. This deubiquitinating enzyme (DUB) has been well-established as a validated oncology target. Using a screening cascade, starting with a 5 million compound high throughput screen towards a 10K library of primary hits, followed by a 2.5K library of secondary hits, an analog was identified with an hUSP21 HTRF IC50 of 11.8 μM. Ultimately, this was optimized towards BAY-805, with an IC50 of 6 nM (~2000x improvement). In a SPR assay, a Kd of 2.2 nM was found, and in a Ub-Rhod hUSP21 study, an IC50 of 2 nM was reported. Also, in a NF-κB reporter assay, an EC50 of 16.6 nM was established.
Reference: https://doi.org/10.1021/acs.jmedchem.2c01933
Compound 2E

Compound 2E: This pyrazolo[3,4-d]pyridazinone was reported a covalent FGFR inhibitor, targeting Cys488 using a canonical acrylamide electrophile (and a 1,4-conjugate addition mechanism). There are 5 isoforms of FGFR (FGFR1, FGFR2, FGFR3, FGFR4, and FGFRL1, which have been extensively studied as oncology targets. Lead optimization towards compound 2E involved structure-based strategies and molecular docking studies (PDB 3TT0). Against FGFR1, FGFR2, FGFR3, an IC50 of 24.1, 13.6 and 52.6 nM was found. In a FGFR2-N549H study an IC50 of 16 nM was reported, and in SNU-16/H716 assays, IC50 values of 1.2/3.2 nM were found. In vivo efficacy was established in 3 FGFR xenograft models, at 200 mg/kg (p.o.), with TGI percentages ranging between 68-83%.
Reference: https://doi.org/10.1021/acs.jmedchem.2c01507
PF-07258669

PF-07258669: This spirocyclic structure out of Pfizer, has been reported as a melanocortin-4 receptor (MC4R) antagonist. MC4R is a GPCR implicated in hyperphagia, anorexia, and cachexia (related to food intake, appetite, weight loss, etc.). Using a screen of 28K analogs, and a structural mechanistic observation of an acyl migration, a series of 1-acylaminopyrrolidines was discovered. Against hMC4R, an IC50 of 13 nM was found, with a Ki of 0.46 nM. In vivo PK studies in rats, at 5 mg/kg (p.o.) revealed a T1/2 of 0.98 hrs, a TMax of 0.5 hrs and an oral bioavailability (% F) of 28%. In relevant animal models of anorexia/cachexia, at 10 mg/kg (BID), a +111% increase of food intake was observed after the administration of this compound.
Reference: https://doi.org/10.1021/acs.jmedchem.2c02012
MH3

MH3: This (R)-3-aminopyrrolidine has been reported as an inhibitor of Melanin Concentrating Hormone (MCH-R1). This GPCR has been also implicated in food intake, weight loss, and appetite circuits. Using a structure-based strategy, and molecular docking, key interactions were predicted with Phe282, Trp248, Asp192, and Gln196. Using a SWISS ADME Boiled-egg model, permeation into the blood-brain-barrier has been predicted. In a radio-ligand binding assay CHO (I125-MCH-R1), a Ki value of 7 nM was reported.
Reference: https://doi.org/10.1016/j.bmcl.2023.129194