In this issue, we bring you 7 recently reported small molecules out of the Journal of Medicinal Chemistry focused on several unique drug targets (beyond strategies focused on well-known kinases, standard enzymes, GPCRs, etc.). This list of targets includes metabolic enzyme MAT2A, heat-shock transcription factor HSF1, chemokine receptor CCR8, mitochondrial RNA polymerase POLRMT, the NLRP3 inflammasome, non-nucleoside reverse transcriptase NNRT, and inhibitor of apoptosis proteins IAP.

Compound 28
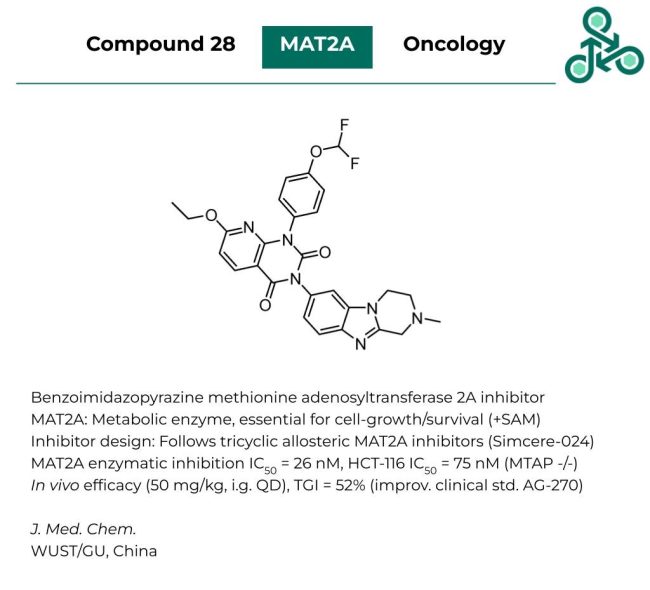
Compound 28: This benzoimidazopyrazine small molecule was recently reported as a methionine adenosyltransferase 2A (MAT2A) inhibitor. MAT2A is a metabolic enzyme, essential for cell-growth, survival, and the production of co-substrate S-Adenosyl methionine (SAM). Inhibitor design is based on a scaffold-hopping strategy from previously known tricyclic allosteric MAT2A inhibitors (Simcere-024). In vitro MAT2A enzymatic inhibition IC50 values of 26 nM were reported, with an IC50 in HCT-116 of 75 nM (MTAP -/-). In vivo efficacy was also established (at 50 mg/kg, i.g., QD), resulting in a TGI value of 52%, which represents an improvement relative to the clinical standard AG-270.
Reference: https://doi.org/10.1021/acs.jmedchem.2c02006
CCT361814
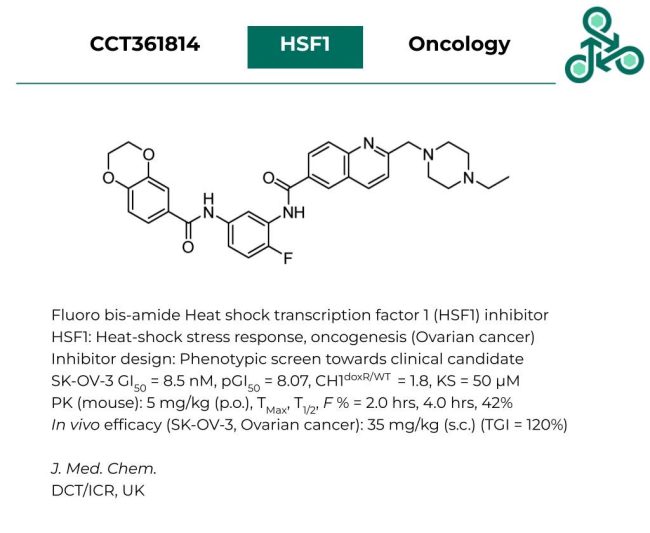
CCT361814: Fluoro bis-amide CCT361814 has been identified as an inhibitor of Heat shock transcription factor 1 (HSF1), a TF implicated in heat-shock stress response, and oncogenesis (specifically within the context of ovarian cancers). Inhibitor design involved a phenotypic screen from Hit-to-Lead, towards a clinical candidate. In SK-OV-3, a GI50 of 8.5 nM was reported, translating into a pGI50 of 8.07. In CH1-doxR/WT, a value of 1.8 was reported, with a KS of 50 µM. In vivo pharmacokinetic analysis in mouse models (5 mg/kg, p.o.) observed a TMax value of 2.0 hrs, a T1/2 of 4.0 hrs, and an oral bioavailability (% F) of 42%. In vivo efficacy was also reported in a SK-OV-3, ovarian cancer tumor model, at 35 mg/kg (s.c.), with a TGI value of 120%.
Reference: https://doi.org/10.1021/acs.jmedchem.3c00156
IPG7236
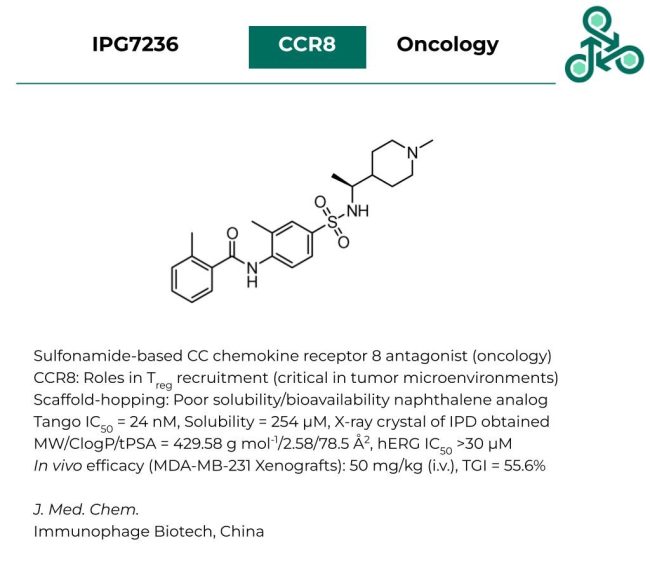
IPG7236: This sulfonamide-based small molecule out of Immunophage Biotech was reported as an antagonist of CC chemokine receptor 8 (CCR8), which has been repeatedly implicated in oncology. CCR8 is involved in regulatory T cells (Treg) recruitment and plays critical roles within the tumor microenvironment of several human cancers. Inhibitor design involved a scaffold-hopping strategy to improve a previously known naphthalene-based lead molecule with poor bioavailability and inadequate solubility. A Tango IC50 of 24 nM was found, with a solubility of 254 µM. As for physicochemical properties, MW, CLogP, and tPSA values of 429.58 g mol-1, 2.58, and 78.5 Å2 were generated with an IC50 >30 µM against hERG. In vivo efficacy was reported in MDA-MB-231 xenografts, 50 mg/kg (i.v.), TGI of 55.6%.
Reference: https://doi.org/10.1021/acs.jmedchem.3c00030
Compound D26
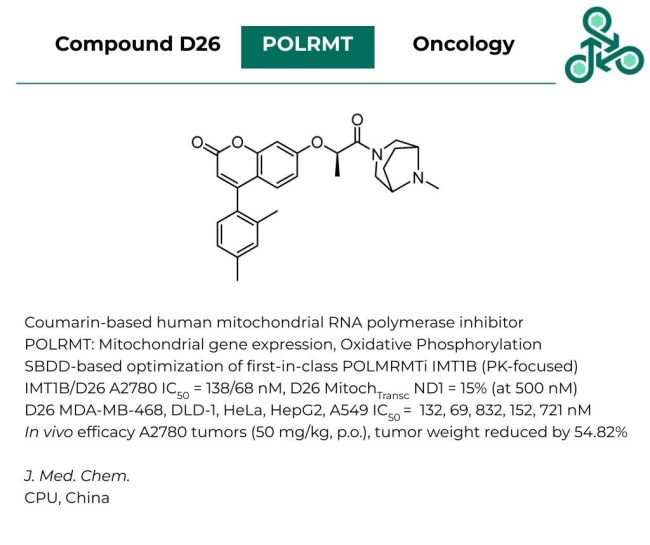
Compound D26: Coumarin-based small molecule D26 has been recently published as an inhibitor of mitochondrial RNA polymerase. POLRMT is an RNA polymerase involved in mitochondrial gene expression and oxidative phosphorylation. Inhibitor design involved a pharmacokinetics-focused structure-based strategy of a previously established first-in-class POLRMT inhibitor, IMT1B. When comparing the two in A2780, an IC50 value of 138 nM, and 68 nM was reported for IMT1B and D26, respectively. As for mitochondrial transcription, D26 returned an ND1 score of 15% (at 500 nM of inhibitor). D26 also observed broad cellular activity in several cell lines, including MDA-MB-468, DLD-1, HeLa, HepG2, and A549, with IC50 values of 132, 69, 832, 152, and 721 nM. In vivo efficacy was also observed in A2780 tumor models, at 50 mg/kg, p.o. with a reduction in tumor weight of 54.82%.
Reference: https://doi.org/10.1021/acs.jmedchem.3c00058
Compound 7n
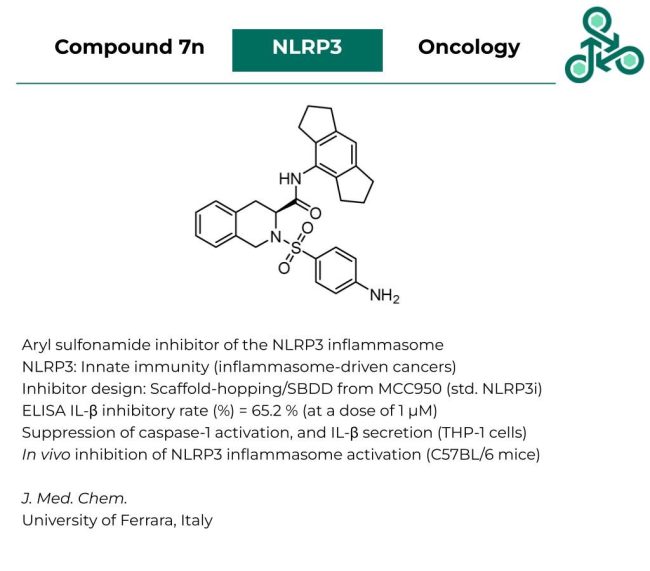
Compound 7n: This aryl sulfonamide from the University of Ferrara in Italy has been reported as inhibitor of the NLRP3 inflammasome. NLRP3 plays critical roles in regulating innate immunity and has been implicated in various inflammasome-driven cancers (e.g. colitis-induced colorectal cancer). Inhibitor design followed a scaffold-hopping and structure-based strategy of a previously known standard NLRP3 inhibitor MCC950. In an ELISA-based assay, the IL-β inhibitory rate (%), at a dose of 1 µM was found to be 65.2%. This compound was also observed to induce the suppression of caspase-1 activation and IL-β secretion (in THP-1 cells). In vivo inhibition of NLRP3 inflammasome activation was also found in C57BL/6 mice.
Reference: https://doi.org/10.1021/acs.jmedchem.3c00175
Compound 8r
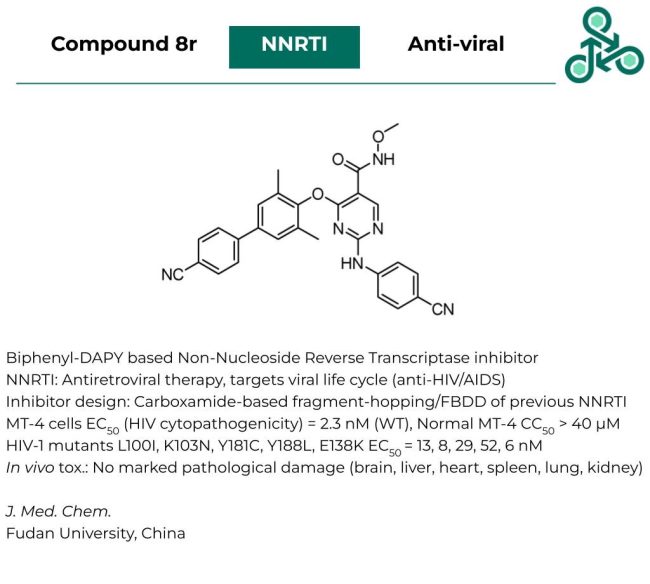
Compound 8r: This DAPY-based biphenyl molecule has been reported as a Non-Nucleoside Reverse Transcriptase inhibitor. NNRTI have been commonly used for Antiretroviral therapy, by targeting several stages of the viral life cycle, with clinical utility as anti-HIV/AIDS therapies. Inhibitor design involved a carboxamide-based fragment-based strategy on a previously established NNRTI. In MT-4 cells, an EC50 (measuring HIV cytopathogenicity), of 2.3 nM was reported (WT), while in normal MT-4 cells, a CC50 > 40 µM was found. Against a series of HIV-1 mutants, L100I, K103N, Y181C, Y188L, and E138K, EC50 values of 13, 8, 29, 52, and 6 nM were observed. In vivo toxicity studies in various critical organs (brain, liver, heart, spleen, lung, and kidney) resulted in no marked pathological damage.
Reference: https://doi.org/10.1021/acs.jmedchem.2c01900
Compound 9
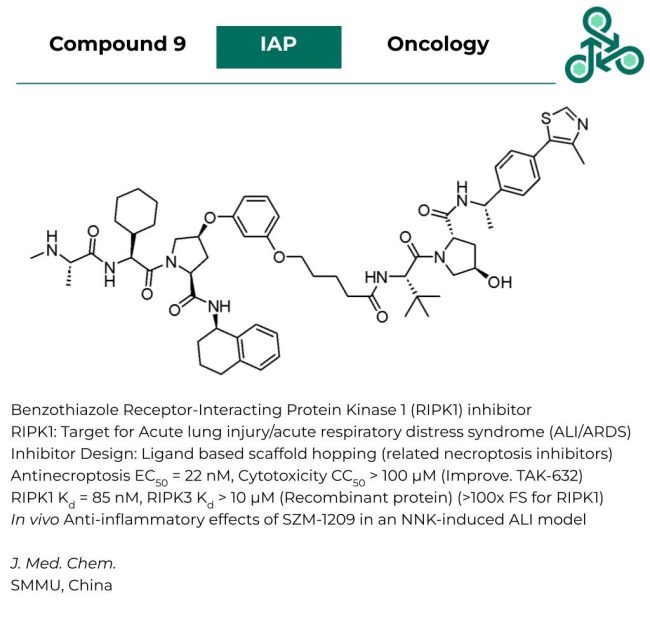
Compound 9: This bi-functional hetero-proteolysis targeting chimera (PROTAC) has been developed as a pan-degrader of inhibitor of apoptosis proteins. Unique E3 ligase cross-talk, results in the potent induction of pan-IAP degradation in MM.1S cells. The degradation profile of compound 9 reveals strong degradation of cIAP1, cIAP2, XIAP, VHL30, CRBN, and IKZF3 levels. In a diaPASEF quantitative LC-MS/MS proteomics study, degradation selectivity was observed for cIAP1, and XIAP (at a dose of 100 nM, for 3 hours). In several cancer cell lines (MM, AML, DLBCL), including NCI-H929, MOLM13, SUDHL6, DB, and K562, IC50 values of 8.5, 2.1, 1.6, 460, and 420 nM were reported.
Reference: https://doi.org/10.1021/acs.jmedchem.2c01817