In this issue of “Small Molecule Highlights” we bring you 5 new small molecules out of recent drug discovery journals. These molecules target a diverse selection of targets, including SARS-CoV-2 3C-like protease, BTK, IKZF1/3, tau, α-synuclein and NS4B. Enjoy!
MK-7845

MK-7845: This SARS-CoV-2 3C-like protease (CLpro) inhibitor developed by Merck took aim at improving the poor pharmacokinetic and toxicity profiles plaguing previous modulators of this target. To address the PK issues, the medicinal chemistry team identified a hepatitis C virus (HCV) protease inhibitor (boceprevir) with weak CLpro activity and greatly improved oral bioavailability as a starting point. To enhance activity, SAR around the glutamine mimic was crucial. This structural motif is common to all CLpro inhibitors and confers selectivity towards the viral protease. Past mimics have utilized a pendant pyrrolidinone substituent to make the necessary H-bond interactions with His164, however, this group adversely affects the solubility and metabolic profile of previous congeners. Employing structural based drug design, the scientists were able to winnow out replacement options to arrive at a unique gem-difluorinated glutamine mimetic with the desired properties. Interestingly, only one of the fluorines was shown to participate in the H-bonding interaction with His164 and fluorine deletion compounds designed to probe this observation were able to substantiate it empirically. Further modification of the parent scaffold, including optimization of the core bicyclic proline and ketoamide warhead, were used to dial in the requisite potency and pharmacokinetic properties. In vitro, MK-7845 was shown to be a potent inhibitor in FRET based CLpro activity assays (IC50 = 8.7 nM) and A549 cellular replicon systems (IC50 = 15 nM). Also, the compound demonstrated similar activity in cellular models of SARS-CoV-2 parental, delta, and omicron infection (EC50 = 444, 351, and 215 nM, respectively). Pharmacokinetic characterization revealed good oral bioavailability in rat and dog (F(%) = 90 and 70, respectively), and a projected human oral bioavailability of 67%. MK-7845 was found to induce CYPs 2B6 and 3A4 at 20 μM. In the case of CYP3A, time-dependant inhibition was observed (Ki/kinact = 110 μM/0.03 min-1), a potential concern for drug-drug interactions at elevated concentrations. Preclinical evaluation in a mouse model of SARS-CoV-2 infection indicated superior control of viral load compared to nirmatrelvir (Pfizer). There was no mention of follow-up clinical studies. In scope, the development of MK-7845 has supplied medicinal chemists with a novel and non-obvious glutamine mimetic (gem-difluorobutyl group). It will be interesting to see how this substituent is deployed in future drug discovery/development efforts.
Reference: https://doi.org/10.1021/acs.jmedchem.3c02248
NX-2127

NX-2127: Scientists at Nurix Therapeutics have developed a dual acting PROTAC/molecular glue targeting both BTK and IKZF1/3. Spurred by results supporting the combination of ibrutinib (BTK covalent inhibitor) and lenalidomide (often used E3 recruiting ligand for PROTACs) for the treatment of diffuse large B cell lymphoma (DLBCL), the combination of the two as a dual targeting agent was too irresistible to pass on. Towards this end, medicinal chemists used structural and in silico design-based approaches to identify a tractable BTK targeting scaffold with the correct exit vector to accommodate an E3 recruiting ligand. Iterative rounds of SAR guided by activity against BTK and IKZF3 eventually led to compound NX-2127. During the structural evolution of NX-2127, one of the key alterations made was deletion of a cyclic urea substituent from the parent scaffold, which helped to increase in vivo potency and limit partitioning in plasma. Characterization of the compound in cells revealed potent BTK degradation in WT and C481S (covalent resistance mutation) harboring TMD8 cells (DC50 = 4.5 nM and 31 nM, respectively), as well as IKZF1 and IKZF3 in primary T cells (DC50 = 57 nM and 36 nM, respectively). Good oral bioavailability in mouse (F(%) = 36) and in vivo BTK degradation was observed across species (%BTK remaining after 24 h = 21/17/10 in mouse/dog/monkey). Pharmacodynamic studies were carried out in xenograft mice containing either BTK WT or C481S transformed TMD8 tumors. Dose-dependant control of tumor volume was observed with NX-2127 with nearly 100% tumor growth inhibition (TGI) occurring at the highest dose tested (90 mg/kg, 5-day, QD, p.o., 25-day trial). Importantly, the compound was able to limit growth in BTK C481S harboring xenografts where no effect was observed with ibrutinib. Follow-up toxicology tests to enable clinical profiling did not identify an issues (AMES was negative; hERG IC50 > 30 μM; no CYP inhibition of major isoforms). Given the agreeable PK and clean toxicology profile, NX-2127 is currently being evaluated patients with B-cell cancers (NCT04830137). As a brief preview of the clinical data, the authors reported sustained degradation of BTK (80%) in CLL patients (100 mg daily, QD, p.o.) enrolled in the trial.
Reference: https://doi.org/10.1021/acs.jmedchem.3c01007
Compound T3

Compound T3: Staying on the topic of dual-targeting degraders, researchers at Sun Yat-sen University recently disclosed their efforts to develop a PROTAC targeting two prominent neurodegenerative disease proteins (NDPs), tau and α-synuclein (α-Syn). As a therapeutic modality, targeting both proteins will likely confer greater benefit as there is a growing body of evidence supporting crosstalk between NDPs. The challenge, finding a ligand/linker/E3 recruiter combination that can effectively degrade both targets selectively whilst maintaining good CNS penetration. For the ligand, choices were relatively meager. The medicinal chemists decided to go with BTA (2-[49-(Methylamino)phenyl]-6-methylbenzothiazole), an analogue of the fluorescent amyloid contrast reagent Thioflavin-T. BTA has previously been shown to bind protein aggregates/oligomers and have good BBB penetration. A relatively limited SAR was conducted around the linker and E3 recruiting substituent, ultimately landing on a PEG4-linked thalidomide as the optimal combination. T3 dose-dependant degraded tau (DC50 = 4.09 μM) and α-Syn (DC50 = 1.57 μM) in manipulated HEK293T cells. Furthermore, degradation of α-Syn was confirmed by proteomics, and immunofluorescence staining coupled with confocal LSM confirmed dose-dependant degradation of tau. T3 was also shown to play a cytoprotective role in a cellular model of protein aggregate induced cell toxicity (SH-SY5Y neuroblastoma cells). Pharmacokinetic characterization revealed low clearance in mouse and rat (CLplasma = 0.1 and 0.12 L/h/kg, respectively), and uptake in the brain. In vivo therapeutic effect was demonstrated in an MPTP induced mouse model of neurotoxic protein aggregation. Specifically, T3 effectively lowered the levels of α-Syn and tau protein aggregates and protected dopaminergic neurons from injury(8 mg/kg, i.v.). A preliminary dose escalation/MTD study in mice to assess in vivo toxicity did not reveal any significant side effect. Taken as a whole, the development of T3 opens a new space for PROTACs in the NDP space, especially in pathologies where more than one target is implicated.
Reference: https://doi.org/10.1021/acs.jmedchem.3c01719
Compound H27
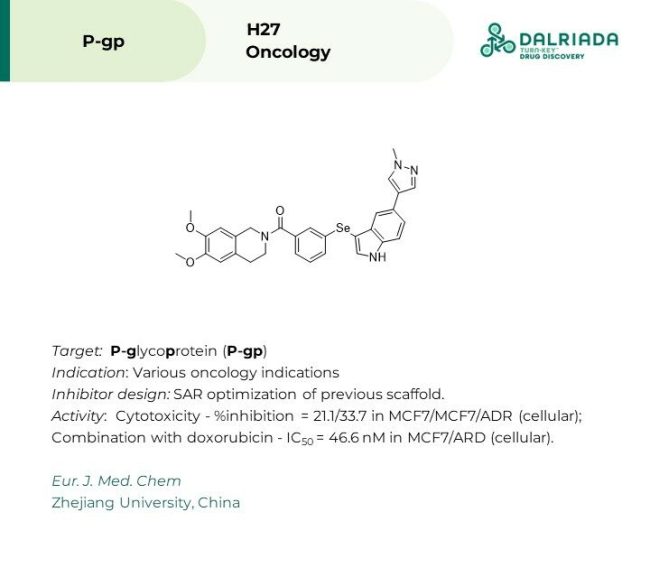
Compound H27: The evolution of multidrug resistance (MDR) in oncology remains a critical challenge in the development of effective chemotherapeutic agents and treatment strategies. While the exact mechanistic underpinnings are still elusive, the overexpression of ABC efflux transporters, particularly P-glycoprotein (P-gp), appears to be one of root causes of MDR. Researchers at the Zhejiang University of Technology have taken an interesting approach to develop novel and non-toxic P-gp inhibitors by incorporating selenium. Commencing from a molecule previously discovered by their group (5m), the medicinal chemists employed a scaffold hopping strategy to replace a problematic furan with a phenyl selenide substituent conferring greater flexibility and lipophilicity. SAR around the groups extending from the phenyl selenide identified a 5-substituted indole as the optimal substituent. Molecular docking studies revealed key interactions between the indole and Phe343 and an H-bond interaction involving the C5-pyrazole and Gln725 of P-gp. In cells, H27 exhibited limited toxicity in WT and MDR MCF7 cells (21.1 and 33.7% inhibition @ 5 μM, respectively). However, in combination with doxorubicin, H27 demonstrated sub-micromolar cytotoxic activity (IC50 = 46.6 nM). Follow up studies in MCF7/ADR cells revealed direct inhibition of P-gp efflux function without modulation of P-gp expression. While H27 does not appear to be a fully optimized compound, it does offer an example of what can be achieved by looking beyond the third row of the periodic table.
Reference: https://doi.org/10.1016/j.ejmech.2024.116207
JNJ-1802

JNJ-1802: Scientists at Janssen recently published details on their lead optimization of a pan-serotype small molecule antiviral for dengue virus. As an aside, dengue related illnesses are some of the fastest growing mosquito-transmitted viral diseases worldwide. Commencing from their previous leads (24a and 28a in https://doi.org/10.1021/acs.jmedchem.3c00403), the team looked to enhance activity across all dengue virus serotypes and the overall pharmacokinetic profile of the scaffold. One of the most interesting alterations was the replacement of a glycol solubilizing group with a methylsulfone substituent. The goal here was to curb glycol associated metabolism and maintain potency. With the sulfone in place, the ortho-methoxyphenyl substituent was essential to maintain the stereochemical configuration of the sole chiral center. Finally, various functional groups and substitution patterns were sampled on the indole group in an effort to further increase potency. A trifluoromethoxy substituent at the C5 position of the indole was found to offer the best balance of target inhibition and metabolic stability. In cellular models of dengue infection, JNJ-1802 exhibited potent activity against all serotypes (EC50 = 0.24/0.057/2.1/11 nM against DENV-1/DENV-2/DENV-3/DENV-4, respectively). Pharmacokinetic characterization revealed good to great oral bioavailability across species (F(%) = 108/58/51/50 in mouse/rat/dog/monkey) coupled with low clearance (CL (mL/min/kg) = 4.97/4.9/6.4/2.45 in mouse/rat/dog/monkey). Clinical enabling safety screens/assays did not flag any cardiotoxicity, genotoxicity, or drug-drug interaction concerns. Pharmacodynamic studies in AG129 mice inoculated with DENV-2 revealed dose-dependant decrease in viral RNA after 3 days. Importantly, viral RNA was undetectable in four of the seven mice treated with the highest dose (60 mg/kg, b.i.d.). Given its potent activity across all serotypes, tractable PK, efficacy in a relevant PD model, and clean toxicity profile, it’s not surprising that JNJ-1802 is progressing to clinical trials.
Reference: https://doi.org/10.1021/acs.jmedchem.3c02336